QUESTION IMAGE
Question
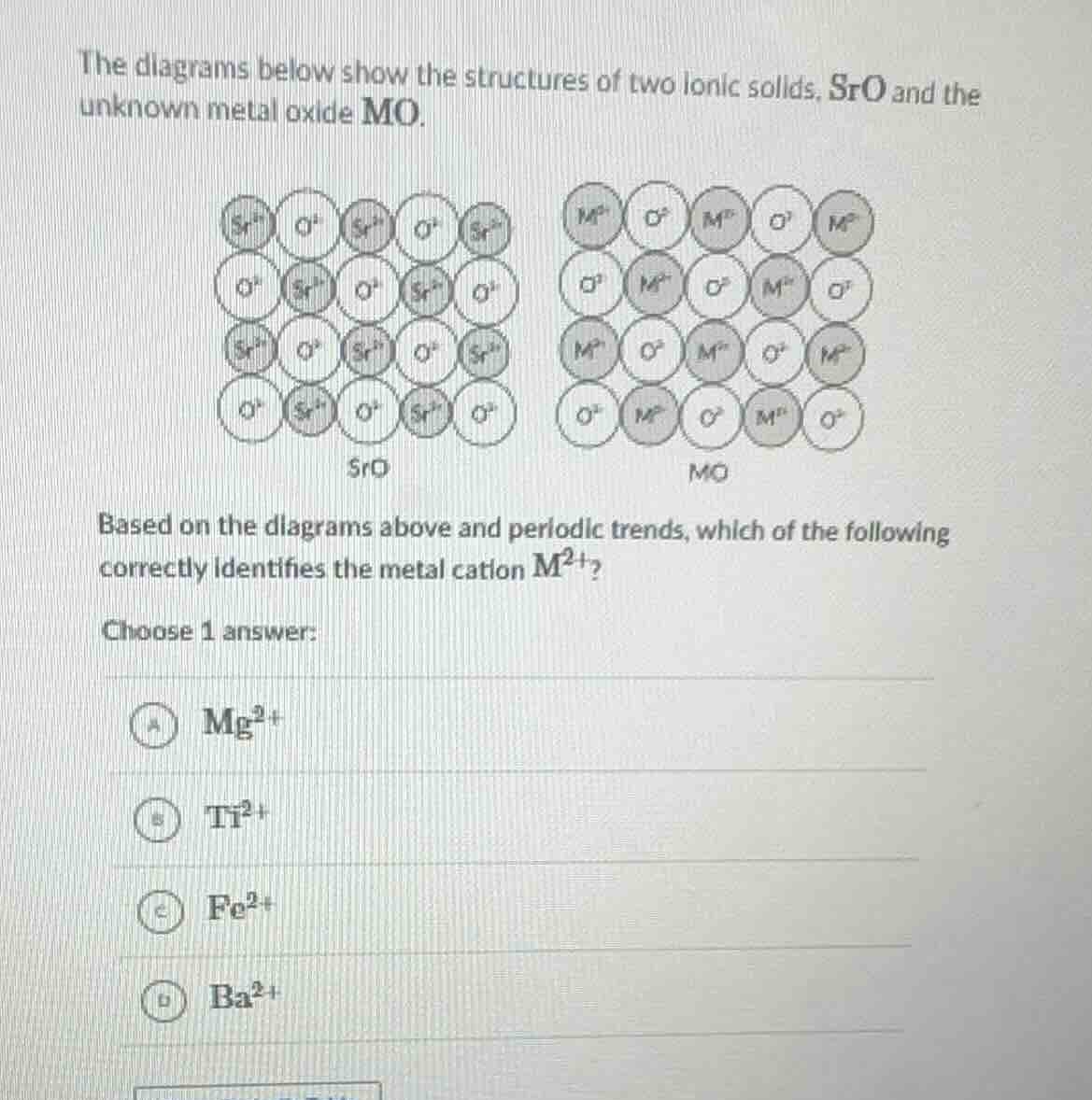
the diagrams below show the structures of two ionic solids, sro and the unknown metal oxide mo. based on the diagrams above and periodic trends, which of the following correctly identifies the metal cation $m^{2+}$? choose 1 answer: a $\ce{mg^{2+}}$ b $\ce{ti^{2+}}$ c $\ce{fe^{2+}}$ d $\ce{ba^{2+}}$
Brief Explanations
- First, analyze the ionic radii trend: In the same group, as the atomic number increases, the ionic radius of metal cations with the same charge increases. Sr is in Group 2. We need to find a cation with a smaller ionic radius than \( \text{Sr}^{2+} \) (since in the MO structure, the \( \text{M}^{2+} \) ions seem to fit more closely or have a different packing, implying a smaller radius than \( \text{Sr}^{2+} \) or appropriate for the structure).
- \( \text{Ba}^{2+} \) is below Sr in Group 2, so its ionic radius is larger than \( \text{Sr}^{2+} \), so D is out.
- \( \text{Ti}^{2+} \) and \( \text{Fe}^{2+} \) are transition metals. Their ionic radii for \( +2 \) charge: \( \text{Ti}^{2+} \) (from Ti in Period 4, Group 4) and \( \text{Fe}^{2+} \) (Period 4, Group 8) have different electron configurations, but we are dealing with Group 2 - like cations? Wait, no, the compound is MO, same as SrO (which is a Group 2 metal oxide, Sr is Group 2). So M should be a Group 2 metal? Wait, no, the options include Mg (Group 2, above Sr), Ti (Group 4), Fe (Group 8), Ba (Group 2, below Sr).
- Wait, the key is ionic radius: In Group 2, from top to bottom (Mg, Ca, Sr, Ba), ionic radius increases. So \( \text{Mg}^{2+} \) has a smaller ionic radius than \( \text{Sr}^{2+} \), \( \text{Ba}^{2+} \) larger. Now, look at the structures: In SrO, the \( \text{Sr}^{2+} \) and \( \text{O}^{2 -} \) are arranged, and in MO, the \( \text{M}^{2+} \) and \( \text{O}^{2 -} \) have a different packing. Wait, maybe the coordination or the size: \( \text{Mg}^{2+} \) is smaller than \( \text{Sr}^{2+} \), so when forming the ionic solid, the arrangement could be different. Also, Ti²⁺ and Fe²⁺ are not Group 2, so their oxides might have different structures (like TiO or FeO, but the structure here is similar to SrO, which is a rock - salt structure? Wait, SrO has a rock - salt structure (like NaCl). MO also seems to have a similar structure. For rock - salt structure, the cations and anions should have appropriate radii. The radius ratio: For rock - salt, the ideal radius ratio \( \frac{r^ +}{r^ -} \) is around 0.414 - 0.732. \( \text{O}^{2 -} \) radius is about 140 pm. \( \text{Sr}^{2+} \) radius is about 113 pm, \( \text{Mg}^{2+} \) is about 72 pm, \( \text{Ba}^{2+} \) is about 135 pm, \( \text{Ti}^{2+} \) is about 86 pm, \( \text{Fe}^{2+} \) is about 77 pm. Wait, but SrO is a rock - salt structure. If M is Mg, which is smaller, the structure could still be rock - salt (since MgO is also rock - salt). Ti²⁺: TiO has a rock - salt structure? Wait, TiO can have rock - salt, but Ti is a transition metal. But the question is about periodic trends. Since Sr is in Group 2, and we need a metal with a smaller ionic radius (since in the diagram, the M²⁺ ions seem to be more closely packed or have a different arrangement, maybe smaller than Sr²⁺). Mg is above Sr in Group 2, so smaller ionic radius. Ba is below, larger. Ti and Fe are transition metals. So the most likely is Mg²⁺, as it is a Group 2 metal with smaller ionic radius than Sr²⁺, fitting the periodic trend of ionic radius in Group 2 (decreases up the group).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. \( \text{Mg}^{2+} \)