QUESTION IMAGE
Question
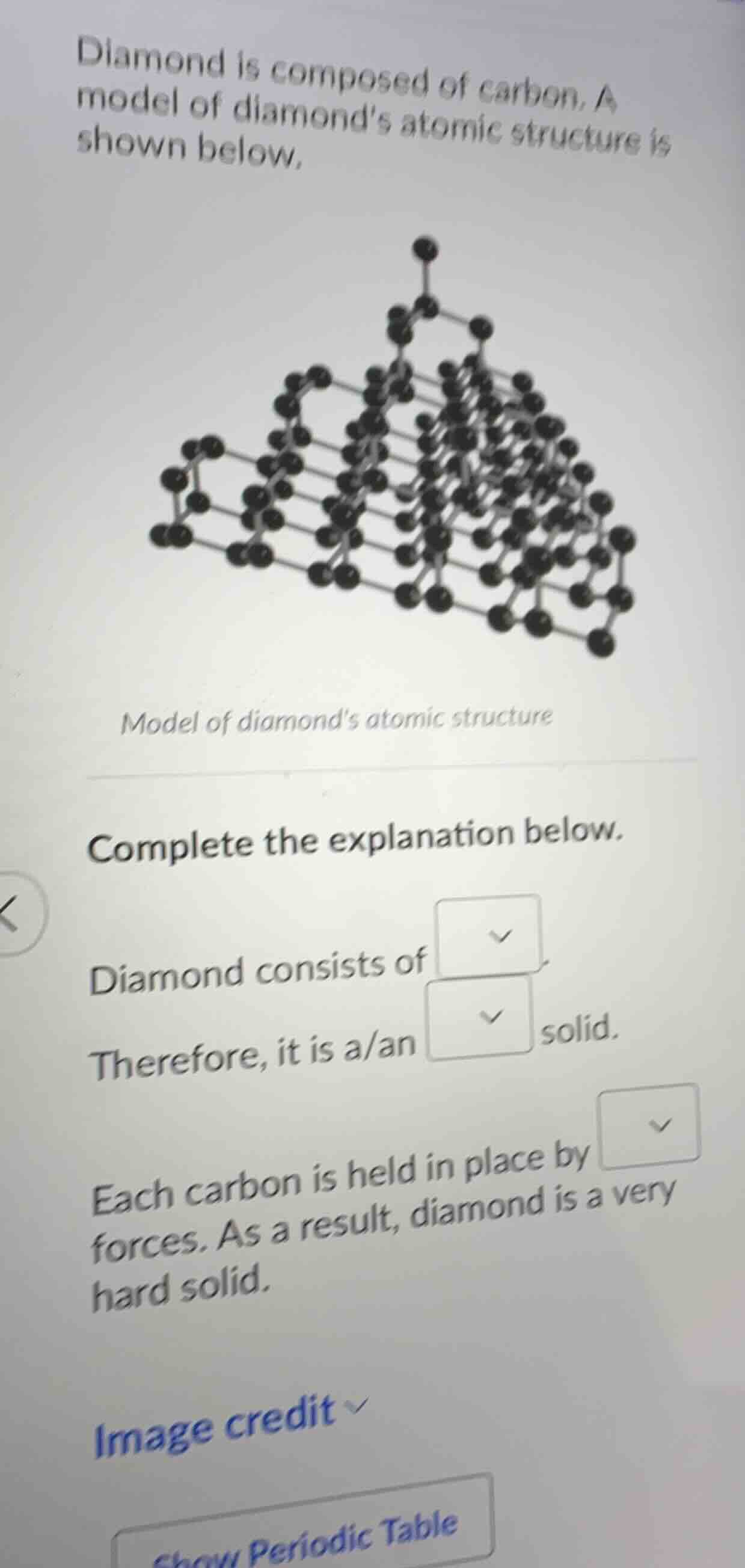
diamond is composed of carbon. a model of diamond’s atomic structure is shown below.
model of diamond’s atomic structure
complete the explanation below.
diamond consists of
therefore, it is a/an solid.
each carbon is held in place by forces. as a result, diamond is a very hard solid.
image credit
show periodic table
Brief Explanations
- Diamond is composed of carbon atoms. In its atomic structure, the carbon atoms are arranged in a repeating, ordered pattern characteristic of a crystal lattice.
- A solid with a highly ordered, repeating atomic structure is classified as a crystalline solid. The strong covalent bonds (the forces holding the carbon atoms in place) in the lattice structure contribute to diamond's hardness. So, the first blank should be "carbon atoms", the second blank "crystalline", and the third blank "covalent" (since each carbon atom is bonded to four others via covalent bonds in diamond's structure).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Diamond consists of \boxed{carbon atoms}, therefore, it is a/an \boxed{crystalline} solid. Each carbon is held in place by \boxed{covalent} forces.