QUESTION IMAGE
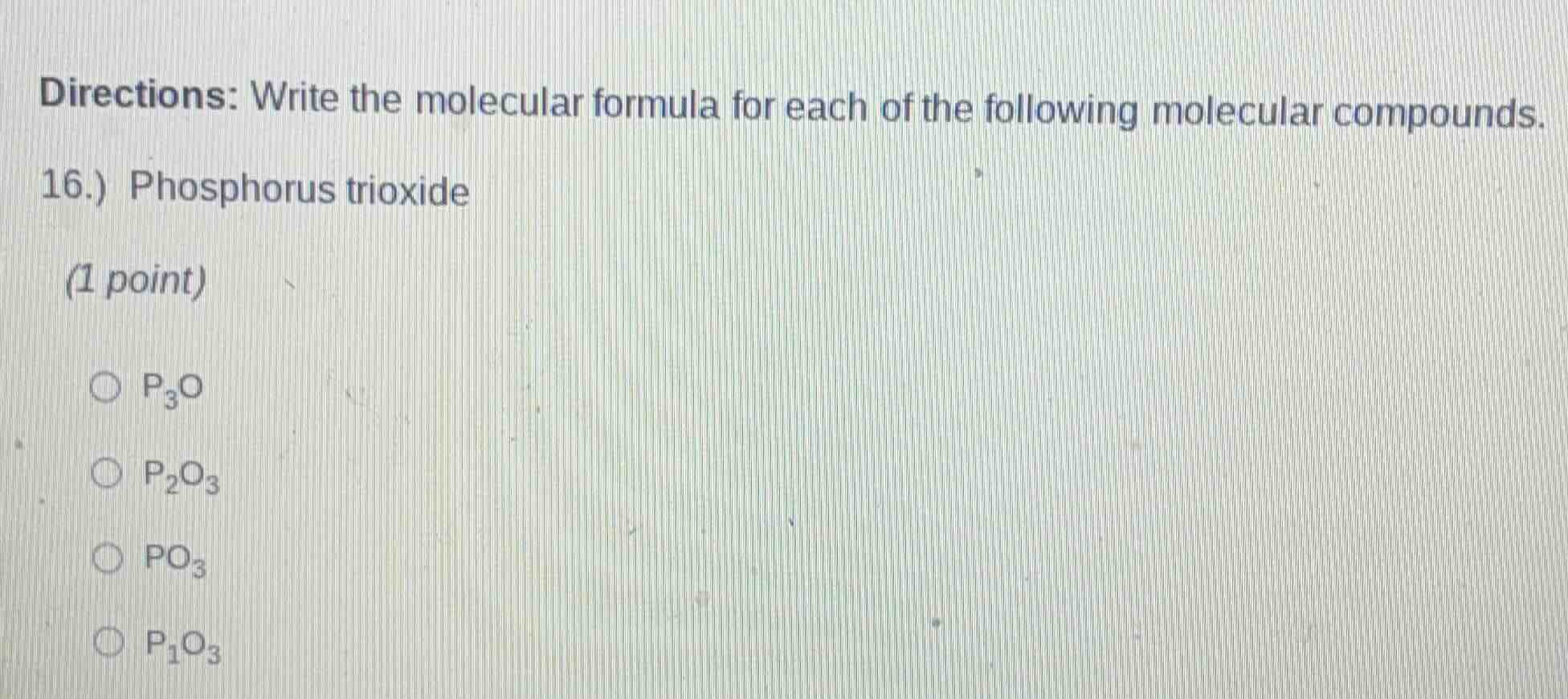
Question
directions: write the molecular formula for each of the following molecular compounds.
16.) phosphorus trioxide
(1 point)
$\boldsymbol{p_3o}$
$\boldsymbol{p_2o_3}$
$\boldsymbol{po_3}$
$\boldsymbol{p_1o_3}$
Step1: Identify element symbols
Phosphorus = $\text{P}$, Oxygen = $\text{O}$
Step2: Use prefixes for subscripts
"Phosphorus" (no prefix) = 2 atoms (standard for phosphorus oxides), "trioxide" = 3 O atoms.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. $\text{P}_2\text{O}_3$