QUESTION IMAGE
Question
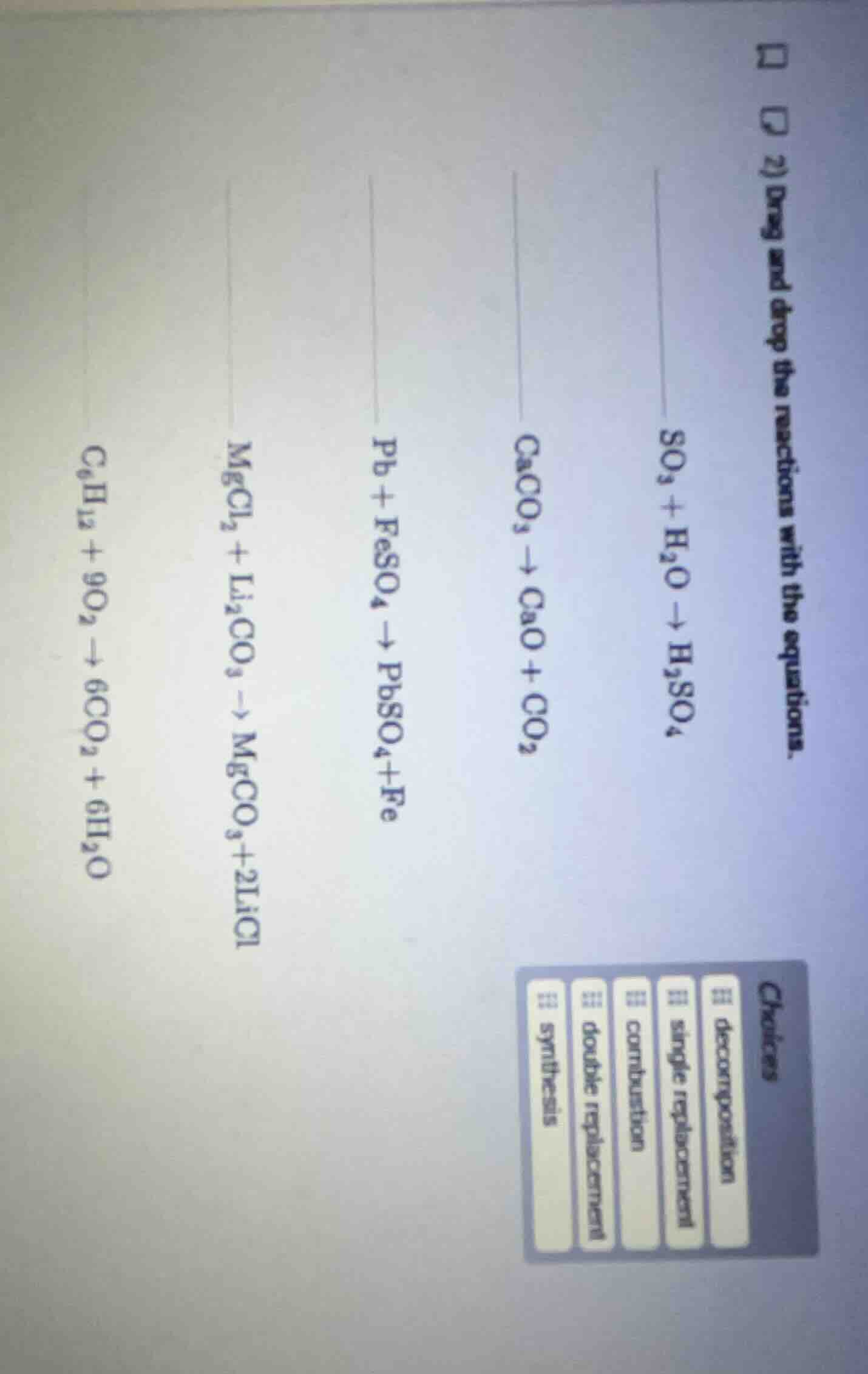
- drag and drop the reactions with the equations. \\(\ce{so3 + h2o -> h2so4}\\) \\(\ce{caco3 -> cao + co2}\\) \\(\ce{pb + feso4 -> pbso4 + fe}\\) \\(\ce{mgcl2 + li2co3 -> mgco3 + 2licl}\\) \\(\ce{c6h12 + 9o2 -> 6co2 + 6h2o}\\) choices: decomposition, single replacement, combination, double replacement, synthesis
- **$\boldsymbol{SO_3 + H_2O
ightarrow H_2SO_4}$**: Two reactants combine into one product, which fits synthesis reaction definition.
- **$\boldsymbol{CaCO_3
ightarrow CaO + CO_2}$**: One reactant breaks into two products, matching decomposition reaction definition.
- **$\boldsymbol{Pb + FeSO_4
ightarrow PbSO_4 + Fe}$**: One element replaces another in a compound, which is a single replacement reaction.
- **$\boldsymbol{MgCl_2 + Li_2CO_3
ightarrow MgCO_3 + 2LiCl}$**: Ions of two compounds swap places, fitting double replacement reaction definition.
- **$\boldsymbol{C_6H_{12} + 9O_2
ightarrow 6CO_2 + 6H_2O}$**: A hydrocarbon reacts with oxygen to produce carbon dioxide and water, which is a combustion reaction.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\boldsymbol{SO_3 + H_2O
ightarrow H_2SO_4}$: synthesis
- $\boldsymbol{CaCO_3
ightarrow CaO + CO_2}$: decomposition
- $\boldsymbol{Pb + FeSO_4
ightarrow PbSO_4 + Fe}$: single replacement
- $\boldsymbol{MgCl_2 + Li_2CO_3
ightarrow MgCO_3 + 2LiCl}$: double replacement
- $\boldsymbol{C_6H_{12} + 9O_2
ightarrow 6CO_2 + 6H_2O}$: combustion