QUESTION IMAGE
Question
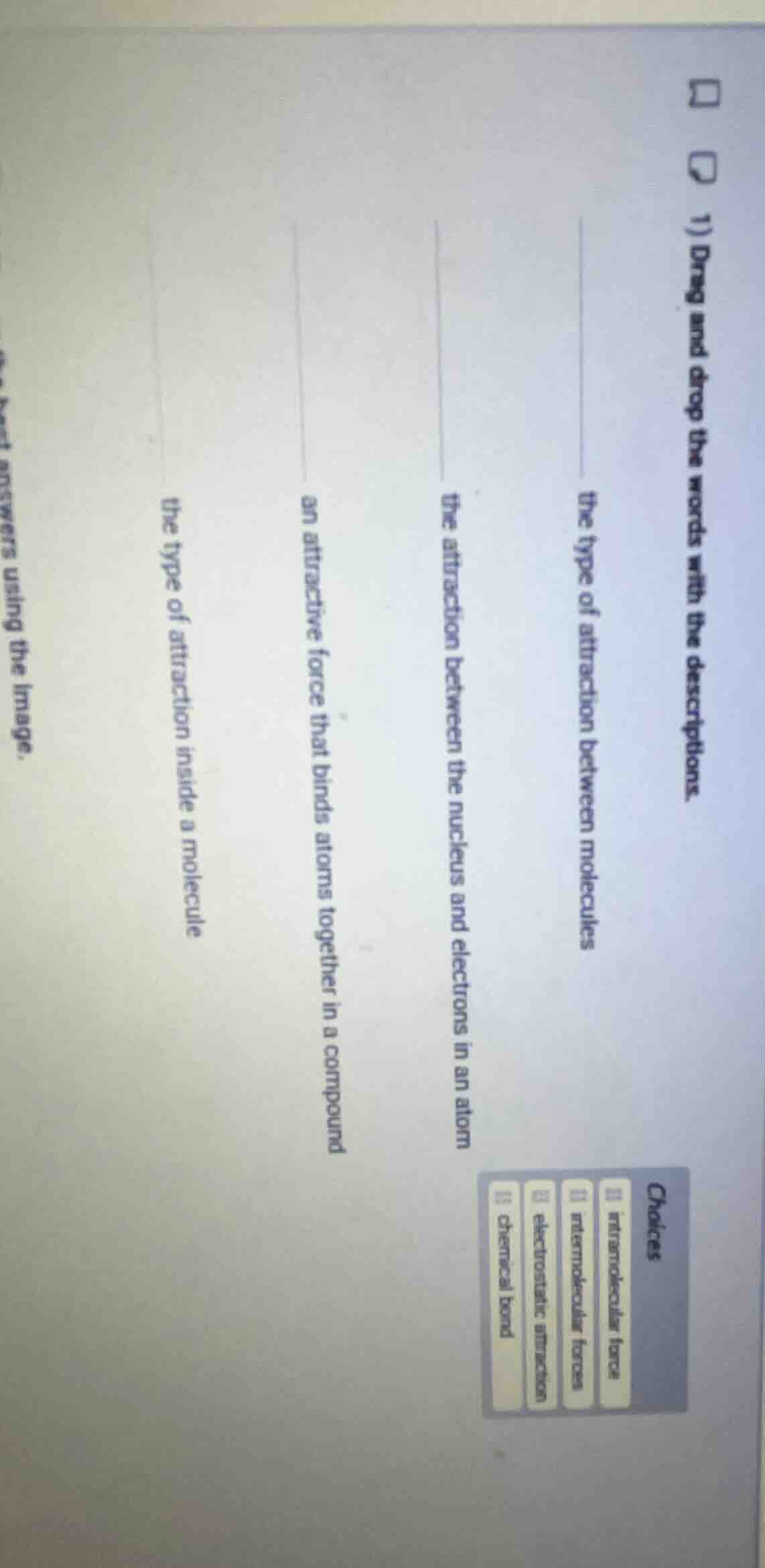
- drag and drop the words with the descriptions. the type of attraction between molecules the attraction between the nucleus and electrons in an atom an attractive force that binds atoms together in a compound the type of attraction inside a molecule choices intramolecular force intermolecular forces electrostatic attraction chemical bond
Brief Explanations
- Intermolecular forces are defined as attractive forces between separate molecules.
- Electrostatic attraction describes the pull between a positively charged atomic nucleus and negatively charged electrons.
- Chemical bonds are the forces that hold atoms together to form compounds.
- Intramolecular forces are the attractive forces that exist within a single molecule.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- intermolecular forces → the type of attraction between molecules
- electrostatic attraction → the attraction between the nucleus and electrons in an atom
- chemical bond → an attractive force that binds atoms together in a compound
- intramolecular force → the type of attraction inside a molecule