QUESTION IMAGE
Question
drag the tiles to the correct boxes to complete the pairs.
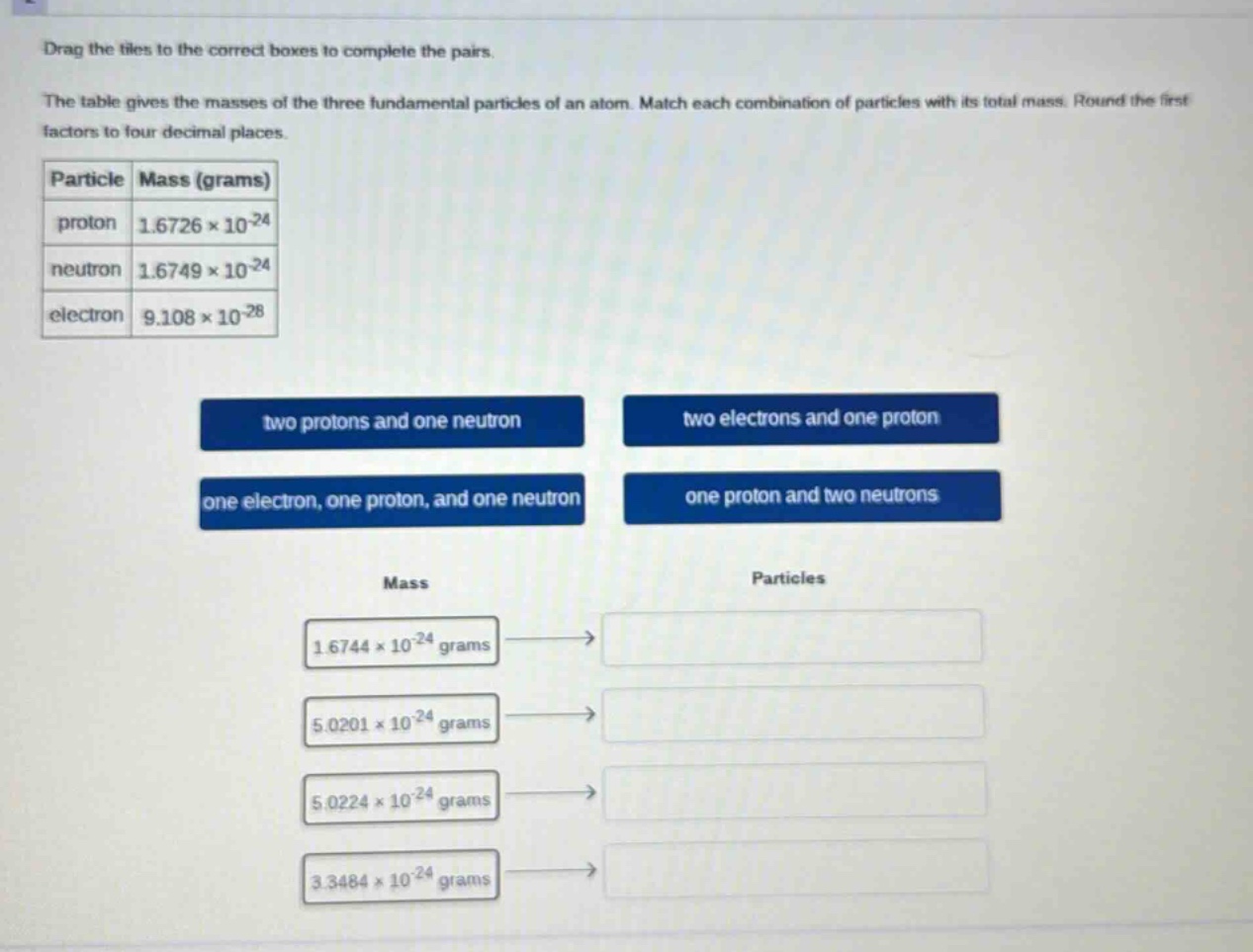
the table gives the masses of the three fundamental particles of an atom. match each combination of particles with its total mass. round the first factors to four decimal places.
particle mass (grams)
proton $1.6726 \times 10^{-24}$
neutron $1.6749 \times 10^{-24}$
electron $9.108 \times 10^{-28}$
two protons and one neutron
two electrons and one proton
one electron, one proton, and one neutron
one proton and two neutrons
mass
particles
$1.6744 \times 10^{-24}$ grams
$5.0201 \times 10^{-24}$ grams
$5.0224 \times 10^{-24}$ grams
$3.3484 \times 10^{-24}$ grams
Step1: Calculate mass of 2 electrons +1 proton
First, convert electron mass to $10^{-24}$: $9.108 \times 10^{-28} = 0.0009108 \times 10^{-24}$.
Total mass: $2\times0.0009108 \times 10^{-24} + 1.6726 \times 10^{-24} = (0.0018216 + 1.6726) \times 10^{-24} = 1.6744216 \times 10^{-24} \approx 1.6744 \times 10^{-24}$ grams
Step2: Calculate mass of 1 proton +2 neutrons
Total mass: $1.6726 \times 10^{-24} + 2\times1.6749 \times 10^{-24} = (1.6726 + 3.3498) \times 10^{-24} = 5.0224 \times 10^{-24}$ grams
Step3: Calculate mass of 2 protons +1 neutron
Total mass: $2\times1.6726 \times 10^{-24} + 1.6749 \times 10^{-24} = (3.3452 + 1.6749) \times 10^{-24} = 5.0201 \times 10^{-24}$ grams
Step4: Calculate mass of 1e,1p,1n
Total mass: $0.0009108 \times 10^{-24} + 1.6726 \times 10^{-24} + 1.6749 \times 10^{-24} = (0.0009108 + 1.6726 + 1.6749) \times 10^{-24} = 3.3484108 \times 10^{-24} \approx 3.3484 \times 10^{-24}$ grams
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$1.6744 \times 10^{-24}$ grams $
ightarrow$ two electrons and one proton
$5.0201 \times 10^{-24}$ grams $
ightarrow$ two protons and one neutron
$5.0224 \times 10^{-24}$ grams $
ightarrow$ one proton and two neutrons
$3.3484 \times 10^{-24}$ grams $
ightarrow$ one electron, one proton, and one neutron