QUESTION IMAGE
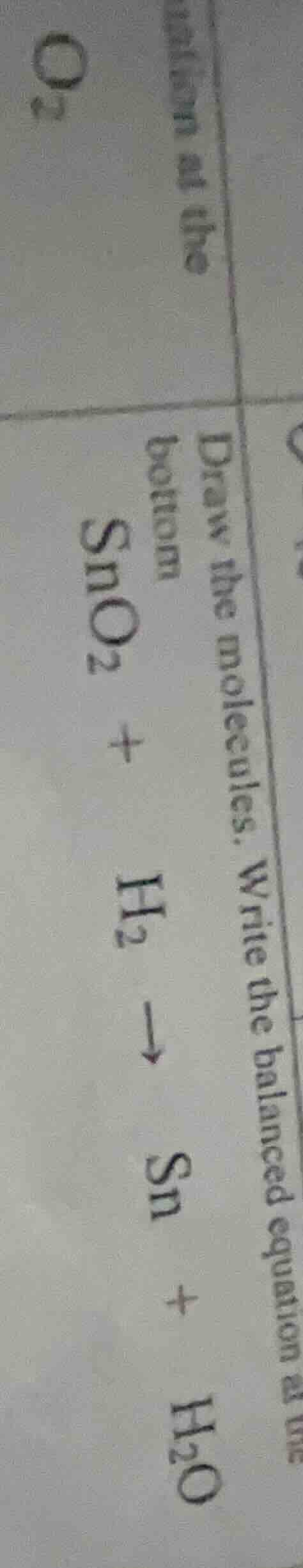
Question
draw the molecules. write the balanced equation at the bottom
$ce{sno_{2} + h_{2} -> sn + h_{2}o}$
Step1: Count atoms on each side
Reactants: Sn=1, O=2, H=2; Products: Sn=1, O=1, H=2
Step2: Balance oxygen atoms
Add coefficient 2 to $\text{H}_2\text{O}$:
$\text{SnO}_2 + \text{H}_2
ightarrow \text{Sn} + 2\text{H}_2\text{O}$
Now: Reactants: Sn=1, O=2, H=2; Products: Sn=1, O=2, H=4
Step3: Balance hydrogen atoms
Add coefficient 2 to $\text{H}_2$:
$\text{SnO}_2 + 2\text{H}_2
ightarrow \text{Sn} + 2\text{H}_2\text{O}$
Now: Reactants: Sn=1, O=2, H=4; Products: Sn=1, O=2, H=4
Step4: Verify all atoms balanced
All elements have equal counts on both sides.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\text{SnO}_2 + 2\text{H}_2
ightarrow \text{Sn} + 2\text{H}_2\text{O}$
(Note: For drawing molecules: $\text{SnO}_2$ is a solid network structure, $\text{H}_2$ is a diatomic molecule ($\text{H-H}$), $\text{Sn}$ is a metallic lattice, $\text{H}_2\text{O}$ is a bent molecule with two $\text{O-H}$ bonds)