QUESTION IMAGE
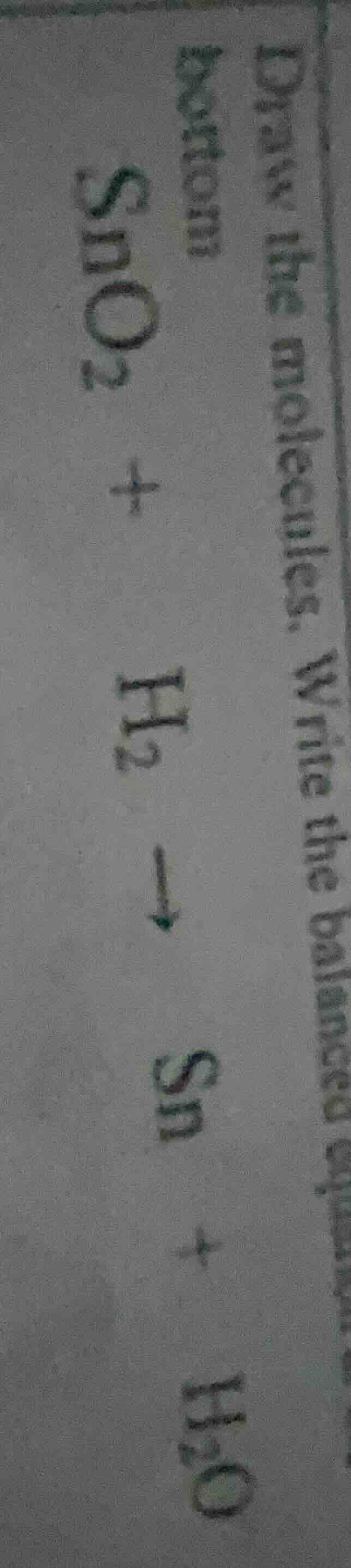
Question
draw the molecules. write the balanced equation
bottom
$ce{sno_{2} + h_{2} -> sn + h_{2}o}$
Step1: List unbalanced atoms
Reactants: Sn=1, O=2, H=2; Products: Sn=1, O=1, H=2
Step2: Balance oxygen atoms
Add coefficient 2 to $\text{H}_2\text{O}$:
$\text{SnO}_2 + \text{H}_2
ightarrow \text{Sn} + 2\text{H}_2\text{O}$
Now: Reactants O=2; Products O=2
Step3: Balance hydrogen atoms
Add coefficient 2 to $\text{H}_2$:
$\text{SnO}_2 + 2\text{H}_2
ightarrow \text{Sn} + 2\text{H}_2\text{O}$
Now: Reactants H=4; Products H=4
Step4: Verify all atoms balanced
Reactants: Sn=1, O=2, H=4; Products: Sn=1, O=2, H=4
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Balanced chemical equation: $\boldsymbol{\text{SnO}_2 + 2\text{H}_2
ightarrow \text{Sn} + 2\text{H}_2\text{O}}$
Molecular Structures (Simplified):
- $\text{SnO}_2$: Tetrahedral structure, Sn at center bonded to 2 double-bonded O atoms (or with resonance/ionic character)
- $\text{H}_2$: Diatomic molecule, single covalent bond between two H atoms: $\text{H-H}$
- $\text{Sn}$: Metallic lattice (single Sn atoms as product)
- $\text{H}_2\text{O}$: Bent (V-shaped) molecule, O at center bonded to two H atoms with two lone pairs on O