QUESTION IMAGE
Question
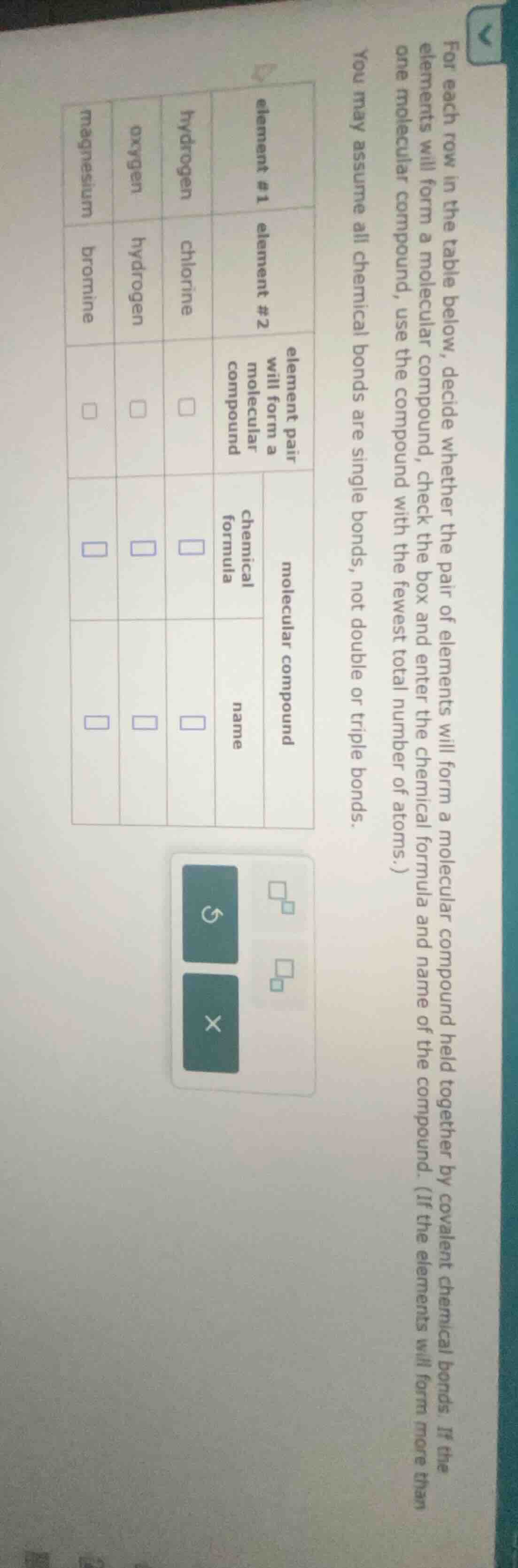
for each row in the table below, decide whether the pair of elements will form a molecular compound, check the box and enter the chemical formula of the compound. (if the elements will form more than one molecular compound, use the compound with the fewest total number of atoms.) you may assume all chemical bonds are single bonds, not double or triple bonds.
| element #1 | element #2 | element pair will form a molecular compound | molecular compound chemical formula | molecular compound name |
|---|---|---|---|---|
| oxygen | hydrogen | $square$ | $square$ | $square$ |
| magnesium | bromine | $square$ | $square$ | $square$ |
Response
To solve this, we analyze each element pair:
Row 1: Hydrogen (H) & Chlorine (Cl)
- Molecular Compound? Yes (both nonmetals, form covalent bonds).
- Formula (fewest atoms): $HCl$ (1 H, 1 Cl; total atoms = 2).
- Name: Hydrogen chloride (or hydrochloric acid, but as a molecular compound, "hydrogen chloride" is precise).
Row 2: Oxygen (O) & Hydrogen (H)
- Molecular Compound? Yes (both nonmetals, form covalent bonds).
- Formula (fewest atoms): $H_2O$ (2 H, 1 O; total atoms = 3).
- Name: Dihydrogen monoxide (or water, but "dihydrogen monoxide" is the systematic name).
Row 3: Magnesium (Mg) & Bromine (Br)
- Molecular Compound? No (Mg is a metal, Br is a nonmetal; they form an ionic compound, not molecular).
Filling the Table:
| Element #1 | Element #2 | Element pair will form a molecular compound? | Molecular compound formula | Molecular compound name |
|---|---|---|---|---|
| Oxygen | Hydrogen | $\boldsymbol{\checkmark}$ | $H_2O$ | Dihydrogen monoxide |
| Magnesium | Bromine | $\boldsymbol{\times}$ | – | – |
Key Notes:
- Molecular compounds form between nonmetals (covalent bonding).
- Ionic compounds (metal + nonmetal) are not molecular, so we mark "×" for Mg/Br.
- For formulas, use the smallest whole-number ratio (oxidation states: H=+1, Cl=-1; O=-2, H=+1).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this, we analyze each element pair:
Row 1: Hydrogen (H) & Chlorine (Cl)
- Molecular Compound? Yes (both nonmetals, form covalent bonds).
- Formula (fewest atoms): $HCl$ (1 H, 1 Cl; total atoms = 2).
- Name: Hydrogen chloride (or hydrochloric acid, but as a molecular compound, "hydrogen chloride" is precise).
Row 2: Oxygen (O) & Hydrogen (H)
- Molecular Compound? Yes (both nonmetals, form covalent bonds).
- Formula (fewest atoms): $H_2O$ (2 H, 1 O; total atoms = 3).
- Name: Dihydrogen monoxide (or water, but "dihydrogen monoxide" is the systematic name).
Row 3: Magnesium (Mg) & Bromine (Br)
- Molecular Compound? No (Mg is a metal, Br is a nonmetal; they form an ionic compound, not molecular).
Filling the Table:
| Element #1 | Element #2 | Element pair will form a molecular compound? | Molecular compound formula | Molecular compound name |
|---|---|---|---|---|
| Oxygen | Hydrogen | $\boldsymbol{\checkmark}$ | $H_2O$ | Dihydrogen monoxide |
| Magnesium | Bromine | $\boldsymbol{\times}$ | – | – |
Key Notes:
- Molecular compounds form between nonmetals (covalent bonding).
- Ionic compounds (metal + nonmetal) are not molecular, so we mark "×" for Mg/Br.
- For formulas, use the smallest whole-number ratio (oxidation states: H=+1, Cl=-1; O=-2, H=+1).