QUESTION IMAGE
Question
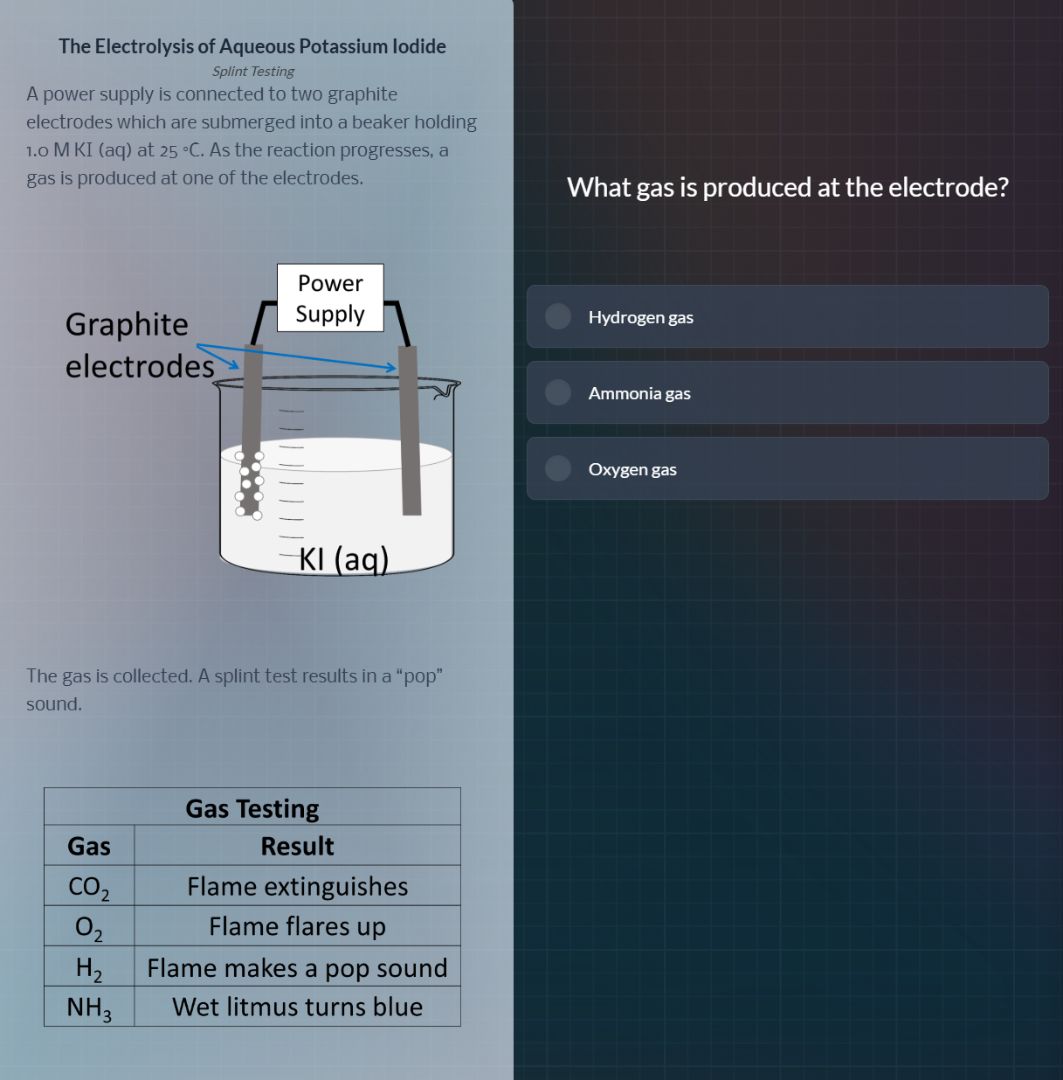
the electrolysis of aqueous potassium iodide
splint testing
a power supply is connected to two graphite
electrodes which are submerged into a beaker holding
1.0 m ki (aq) at 25 °c. as the reaction progresses, a
gas is produced at one of the electrodes.
the gas is collected. a splint test results in a \pop\
sound.
gas testing
gas\tresult
co₂\tflame extinguishes
o₂\tflame flares up
h₂\tflame makes a pop sound
nh₃\twet litmus turns blue
what gas is produced at the electrode?
hydrogen gas
ammonia gas
oxygen gas
- First, analyze the electrolysis of aqueous KI. The ions present are \(K^+\), \(I^-\), \(H^+\), and \(OH^-\) (from water).
- At the cathode (reduction), the possible reactions are the reduction of \(H^+\) (from water) to \(H_2\) or \(K^+\) to \(K\). Since \(K\) is very reactive, \(H^+\) is reduced: \(2H_2O + 2e^-
ightarrow H_2 + 2OH^-\). At the anode (oxidation), \(I^-\) is oxidized to \(I_2\) (but the question is about the gas produced, so focus on cathode).
- The splint test gave a "pop" sound. From the gas testing table, \(H_2\) gives a pop sound, \(O_2\) flares the flame, \(NH_3\) affects litmus, and there's no \(NH_3\) source here. So the gas is \(H_2\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Hydrogen gas