QUESTION IMAGE
Question
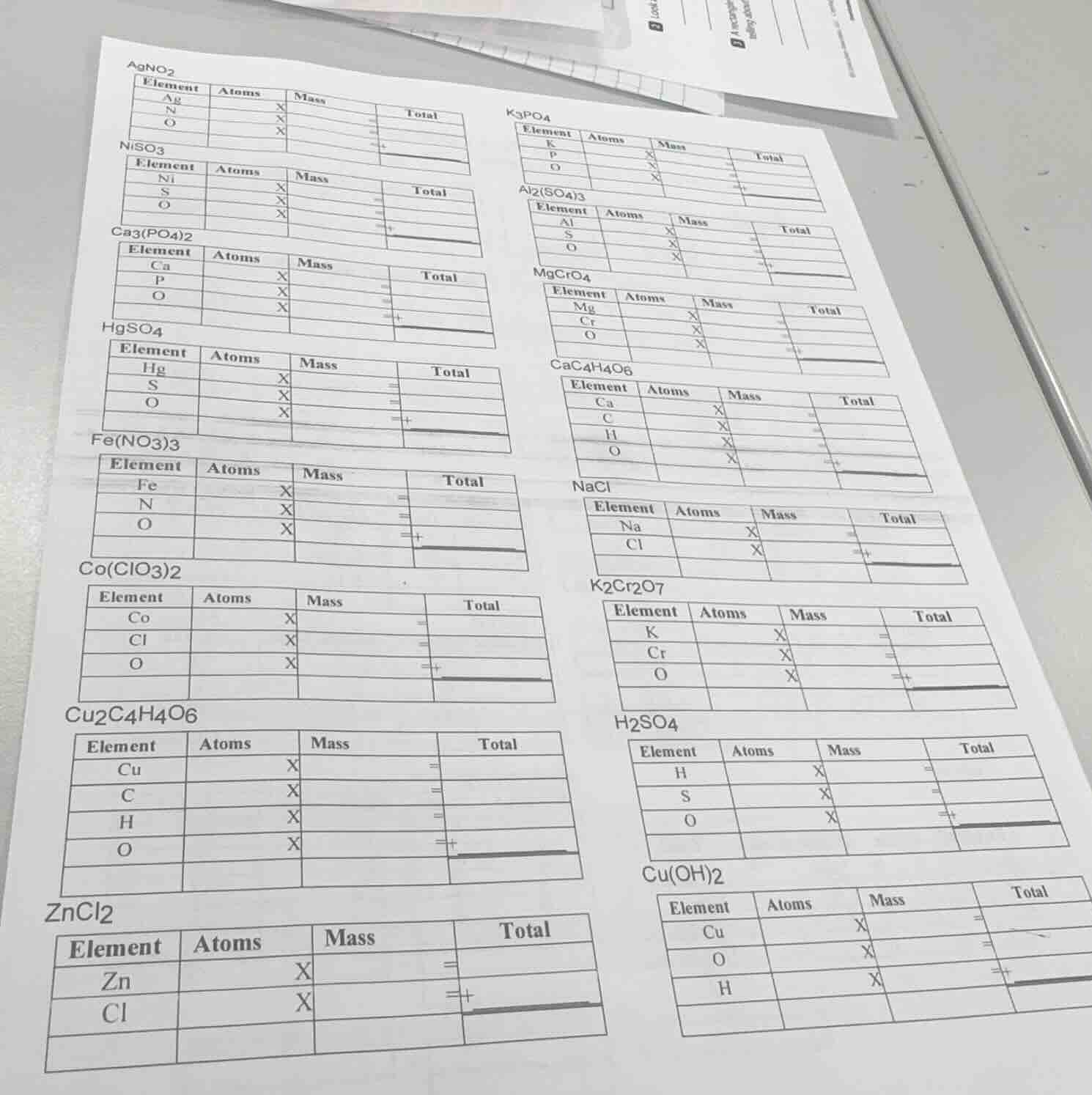
- $\boldsymbol{agno_2}$
| element | atoms | mass | total |
|---|---|---|---|
| n | x | = | |
| o | x | = | + |
- $\boldsymbol{niso_3}$
| element | atoms | mass | total |
|---|---|---|---|
| s | x | = | |
| o | x | = | + |
- $\boldsymbol{ca_3(po_4)_2}$
| element | atoms | mass | total |
|---|---|---|---|
| p | x | = | |
| o | x | = | + |
- $\boldsymbol{hgso_4}$
| element | atoms | mass | total |
|---|---|---|---|
| s | x | = | |
| o | x | = | + |
- $\boldsymbol{fe(no_3)_3}$
| element | atoms | mass | total |
|---|---|---|---|
| n | x | = | |
| o | x | = | + |
- $\boldsymbol{co(clo_3)_2}$
| element | atoms | mass | total |
|---|---|---|---|
| cl | x | = | |
| o | x | = | + |
- $\boldsymbol{cu_2c_4h_4o_6}$
| element | atoms | mass | total |
|---|---|---|---|
| c | x | = | |
| h | x | = | |
| o | x | = | + |
- $\boldsymbol{zncl_2}$
| element | atoms | mass | total |
|---|---|---|---|
| cl | x | = | + |
- $\boldsymbol{k_3po_4}$
| element | atoms | mass | total |
|---|---|---|---|
| p | x | = | |
| o | x | = | + |
- $\boldsymbol{al_2(so_4)_3}$
| element | atoms | mass | total |
|---|---|---|---|
| s | x | = | |
| o | x | = | + |
- $\boldsymbol{mgcro_4}$
| element | atoms | mass | total |
|---|---|---|---|
| cr | x | = | |
| o | x | = | + |
- $\boldsymbol{cac_4h_4o_6}$
| element | atoms | mass | total |
|---|---|---|---|
| c | x | = | |
| h | x | = | |
| o | x | = | + |
- $\boldsymbol{nacl}$
| element | atoms | mass | total |
|---|---|---|---|
| cl | x | = | + |
- $\boldsymbol{k_2cr_2o_7}$
| element | atoms | mass | total |
|---|---|---|---|
| cr | x | = | |
| o | x | = | + |
- $\boldsymbol{h_2so_4}$
| element | atoms | mass | total |
|---|---|---|---|
| s | x | = | |
| o | x | = | + |
- $\boldsymbol{cu(oh)_2}$
| element | atoms | mass | total |
|---|---|---|---|
| o | x | = | |
| h | x | = | + |
Step1: Find atomic counts per compound
Use subscripts to count atoms:
- $\text{AgNO}_3$: $\text{Ag}=1$, $\text{N}=1$, $\text{O}=3$
- $\text{NiSO}_3$: $\text{Ni}=1$, $\text{S}=1$, $\text{O}=3$
- $\text{Ca}_3(\text{PO}_4)_2$: $\text{Ca}=3$, $\text{P}=2$, $\text{O}=8$
- $\text{HgSO}_4$: $\text{Hg}=1$, $\text{S}=1$, $\text{O}=4$
- $\text{Fe(NO}_3\text{)}_3$: $\text{Fe}=1$, $\text{N}=3$, $\text{O}=9$
- $\text{Co(ClO}_3\text{)}_2$: $\text{Co}=1$, $\text{Cl}=2$, $\text{O}=6$
- $\text{Cu}_2\text{C}_4\text{H}_4\text{O}_6$: $\text{Cu}=2$, $\text{C}=4$, $\text{H}=4$, $\text{O}=6$
- $\text{ZnCl}_2$: $\text{Zn}=1$, $\text{Cl}=2$
- $\text{K}_3\text{PO}_4$: $\text{K}=3$, $\text{P}=1$, $\text{O}=4$
- $\text{Al}_2(\text{SO}_4\text{)}_3$: $\text{Al}=2$, $\text{S}=3$, $\text{O}=12$
- $\text{MgCrO}_4$: $\text{Mg}=1$, $\text{Cr}=1$, $\text{O}=4$
- $\text{CaC}_4\text{H}_4\text{O}_6$: $\text{Ca}=1$, $\text{C}=4$, $\text{H}=4$, $\text{O}=6$
- $\text{NaCl}$: $\text{Na}=1$, $\text{Cl}=1$
- $\text{K}_2\text{Cr}_2\text{O}_7$: $\text{K}=2$, $\text{Cr}=2$, $\text{O}=7$
- $\text{H}_2\text{SO}_4$: $\text{H}=2$, $\text{S}=1$, $\text{O}=4$
- $\text{Cu(OH)}_2$: $\text{Cu}=1$, $\text{O}=2$, $\text{H}=2$
Step2: Get atomic masses (g/mol)
Use standard atomic masses:
$\text{Ag}=107.87$, $\text{N}=14.01$, $\text{O}=16.00$, $\text{Ni}=58.69$, $\text{S}=32.07$, $\text{Ca}=40.08$, $\text{P}=30.97$, $\text{Hg}=200.59$, $\text{Fe}=55.85$, $\text{Co}=58.93$, $\text{Cl}=35.45$, $\text{Cu}=63.55$, $\text{C}=12.01$, $\text{H}=1.008$, $\text{Zn}=65.38$, $\text{K}=39.10$, $\text{Al}=26.98$, $\text{Mg}=24.31$, $\text{Cr}=52.00$, $\text{Na}=22.99$
Step3: Calculate total mass per element
Total = Atoms $\times$ Atomic Mass
Step4: Sum for molar mass
Molar mass = Sum of all element totals
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\text{AgNO}_3$
| Element | Atoms | Mass (g/mol) | Total (g/mol) |
|---|---|---|---|
| N | 1 | 14.01 | $1 \times 14.01 = 14.01$ |
| O | 3 | 16.00 | $3 \times 16.00 = 48.00$ |
| Total Molar Mass | --- | --- | $107.87 + 14.01 + 48.00 = 169.88$ |
$\text{NiSO}_3$
| Element | Atoms | Mass (g/mol) | Total (g/mol) |
|---|---|---|---|
| S | 1 | 32.07 | $1 \times 32.07 = 32.07$ |
| O | 3 | 16.00 | $3 \times 16.00 = 48.00$ |
| Total Molar Mass | --- | --- | $58.69 + 32.07 + 48.00 = 138.76$ |
$\text{Ca}_3(\text{PO}_4)_2$
| Element | Atoms | Mass (g/mol) | Total (g/mol) |
|---|---|---|---|
| P | 2 | 30.97 | $2 \times 30.97 = 61.94$ |
| O | 8 | 16.00 | $8 \times 16.00 = 128.00$ |
| Total Molar Mass | --- | --- | $120.24 + 61.94 + 128.00 = 310.18$ |
$\text{HgSO}_4$
| Element | Atoms | Mass (g/mol) | Total (g/mol) |
|---|---|---|---|
| S | 1 | 32.07 | $1 \times 32.07 = 32.07$ |
| O | 4 | 16.00 | $4 \times 16.00 = 64.00$ |
| Total Molar Mass | --- | --- | $200.59 + 32.07 + 64.00 = 296.66$ |
$\text{Fe(NO}_3\text{)}_3$
| Element | Atoms | Mass (g/mol) | Total (g/mol) |
|---|---|---|---|
| N | 3 | 14.01 | $3 \times 14.01 = 42.03$ |
| O | 9 | 16.00 | $9 \times 16.00 = 144.00$ |
| Total Molar Mass | --- | --- | $55.85 + 42.03 + 144.00 = 241.88$ |
$\text{Co(ClO}_3\text{)}_2$
| Element | Atoms | Mass (g/mol) | Total (g/mol) |
|---|---|---|---|
| Cl | 2 | 35.45 | $2 \times 35.45 = 70.90$ |
| O | 6 | 16.00 | $6 \times 16.00 = 96.00$ |
| Total Molar Mass | --- | --- | $58.93 + 70.90 + 96.00 = 225.83$ |
$\text{Cu}_2\text{C}_4\text{H}_4\text{O}_6$
| Element | Atoms | Mass (g/mol) | Total (g/mol) |
|---|---|---|---|
| C | 4 | 12.01 | $4 \times 12.01 = 48.04$ |
| H | 4 | 1.008 | $4 \times 1.008 = 4.032$ |
| O | 6 | 16.00 | $6 \times 16.00 = 96.00$ |
| Total Molar Mass | --- | --- | $127.10 + 48.04 + 4.032 + 96.00 = 275.17$ |
$\text{ZnCl}_2$
| Element | Atoms | Mass (g/mol) | Total (g/mol) |
|---|---|---|---|
| Cl | 2 | 35.45 | $2 \times 35.45 = 70.90$ |
| Total Molar Mass | --- | --- | $65.38 + 70.90 = 136.28$ |
$\text{K}_3\text{PO}_4$
| Element | Atoms | Mass (g/mol) | Total (g/mol) |
|---|---|---|---|
| P | 1 | 30.97 | $1 \times 30.97 = 30.97$ |
| O | 4 | 16.00 | $4 \times 16.00 = 64.00$ |
| Total Molar Mass | --- | --- | $117.30 + 30.97 + 64.00 = 212.27$ |
$\text{Al}_2(\text{SO}_4\text{)}_3$
| Element | Atoms | Mass (g/mol) | Total (g/mol) |
|---|---|---|---|
| S | 3 | 32.07 | $3 \times 32.07 = 96.21$ |
| O | 12 | 16.00 | $12 \times 16.00 = 192.00$ |
| Total Molar Mass | --- | --- | $53.96 + 96.21 + 192.00 = 342.17$ |
$\text{MgCrO}_4$
| Element | Atoms | Mass (g/mol) | Total (g/mol) |
|---|---|---|---|
| Cr | 1 | 52.00 | $1 \times 52.00 = 52.00$ |
| O | 4 | 16.00 | $4 \times 16.00 = 64.00$ |
| Total Molar Mass | --- | --- | $24.31 + 52.00 + 64.00 = 140.31$ |
$\text{CaC}_4\text{H}_4\text{O}_6$
| Element | Atoms | Mass (g/mol) | Total (g/mol) |
|---|---|---|---|
| C | 4 | 12.01 | $4 \times 12.01 = 48.04$ |
| H | 4 | 1.008 | $4 \times 1.008 = 4.032$ |
| O | 6 | 16.00 | $6 \times 16.00 = 96.00$ |
| Total Molar Mass | --- | --- | $40.08 + 48.04 + 4.032 + 96.00 = 188.15$ |
$\text{NaCl}$
| Element | Atoms | Mass (g/mol) | Total (g/mol) |
|---|---|---|---|
| Cl | 1 | 35.45 | $1 \times 35.45 = 35.45$ |
| Total Molar Mass | --- | --- | $22.99 + 35.45 = 58.44$ |
$\text{K}_2\text{Cr}_2\text{O}_7$
| Element | Atoms | Mass (g/mol) | Total (g/mol) |
|---|---|---|---|
| Cr | 2 | 52.00 | $2 \times 52.00 = 104.00$ |
| O | 7 | 16.00 | $7 \times 16.00 = 112.00$ |
| Total Molar Mass | --- | --- | $78.20 + 104.00 + 112.00 = 294.20$ |
$\text{H}_2\text{SO}_4$
| Element | Atoms | Mass (g/mol) | Total (g/mol) |
|---|---|---|---|
| S | 1 | 32.07 | $1 \times 32.07 = 32.07$ |
| O | 4 | 16.00 | $4 \times 16.00 = 64.00$ |
| Total Molar Mass | --- | --- | $2.016 + 32.07 + 64.00 = 98.09$ |
$\text{Cu(OH)}_2$
| Element | Atoms | Mass (g/mol) | Total (g/mol) |
|---|---|---|---|
| O | 2 | 16.00 | $2 \times 16.00 = 32.00$ |
| H | 2 | 1.008 | $2 \times 1.008 = 2.016$ |
| Total Molar Mass | --- | --- | $63.55 + 32.00 + 2.016 = 97.57$ |