QUESTION IMAGE
Question
the element name.
the -ide name.
oms of each
st element.
multiple-choice question
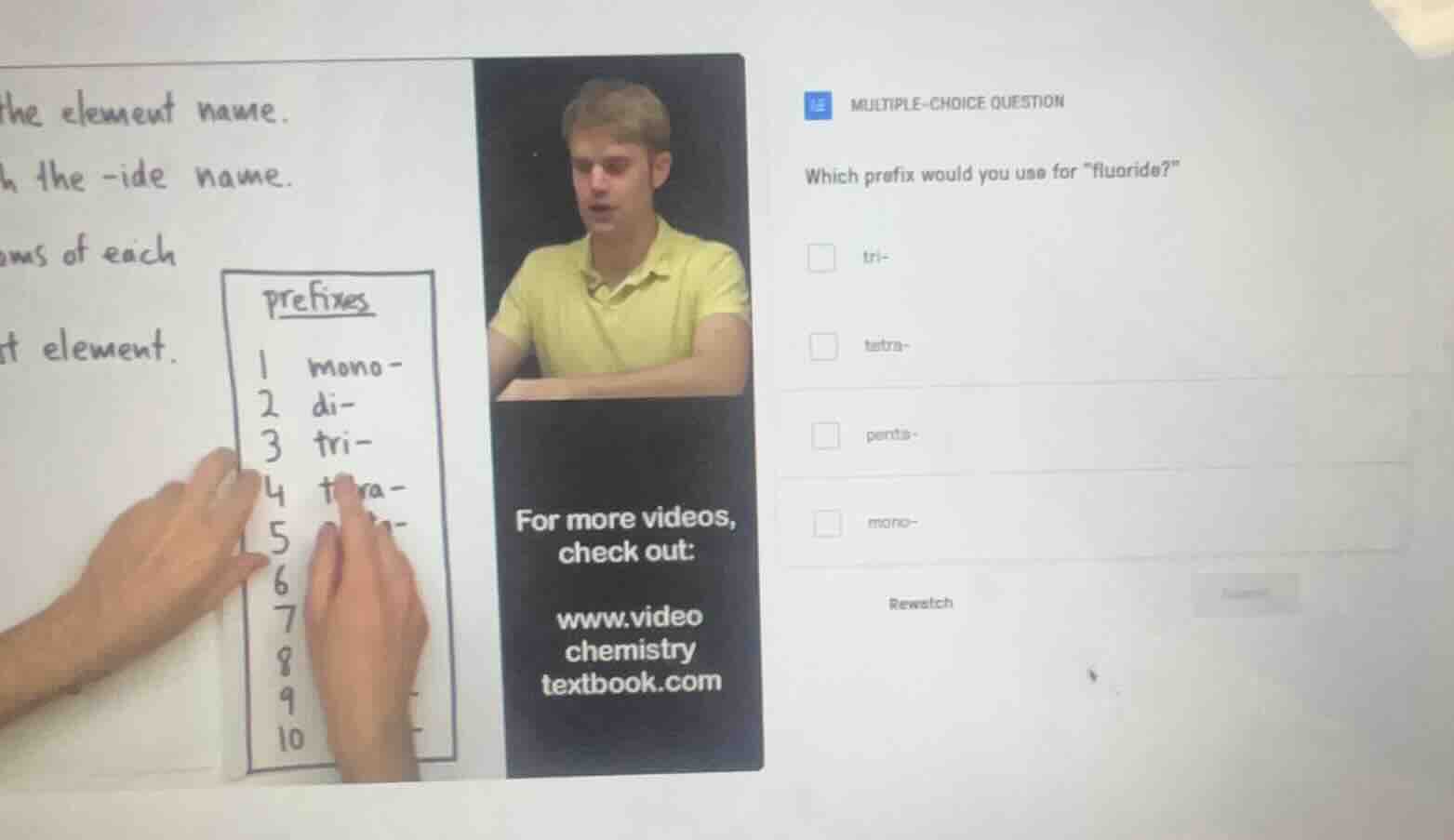
which prefix would you use for \fluoride?\
□ tri-
□ tetra-
□ penta-
□ mono-
rewatch
prefixes
1 mono-
2 di-
3 tri-
4 tetra-
5
6
7
8
9
10
for more videos,
check out:
www.video
chemistry
textbook.com
In chemical nomenclature, when naming binary molecular compounds (like those with fluoride as the anion in some cases, or for molecular fluorine compounds), the prefix "mono - " is used for one atom of the first element (or when there's one fluoride in a context where prefixes apply, though fluoride as an anion in ionic compounds doesn't use prefixes, but in molecular compounds like \( \text{OF}_2 \) is oxygen difluoride, but for \( \text{F}_2 \) it would be diatomic, but if we consider a compound like \( \text{NF}_3 \) is nitrogen trifluoride, but for a single fluoride in a molecular context where the first element has one atom, "mono - " is the prefix for one. Among the options, "mono - " is the correct prefix for one atom (e.g., in \( \text{HF} \) if we were using molecular prefixes, but actually \( \text{HF} \) is ionic, but in molecular compounds like \( \text{OF}_2 \) is oxygen difluoride, \( \text{NF}_3 \) nitrogen trifluoride, \( \text{CF}_4 \) carbon tetrafluoride, \( \text{PF}_5 \) phosphorus pentafluoride. But if we have a compound with one fluoride and one of another element (molecular), like \( \text{XF} \) (where X is a non - metal), it would be X mono - fluoride. The other prefixes: tri - is for three, tetra - for four, penta - for five. So "mono - " is the correct prefix here.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
mono-