QUESTION IMAGE
Question
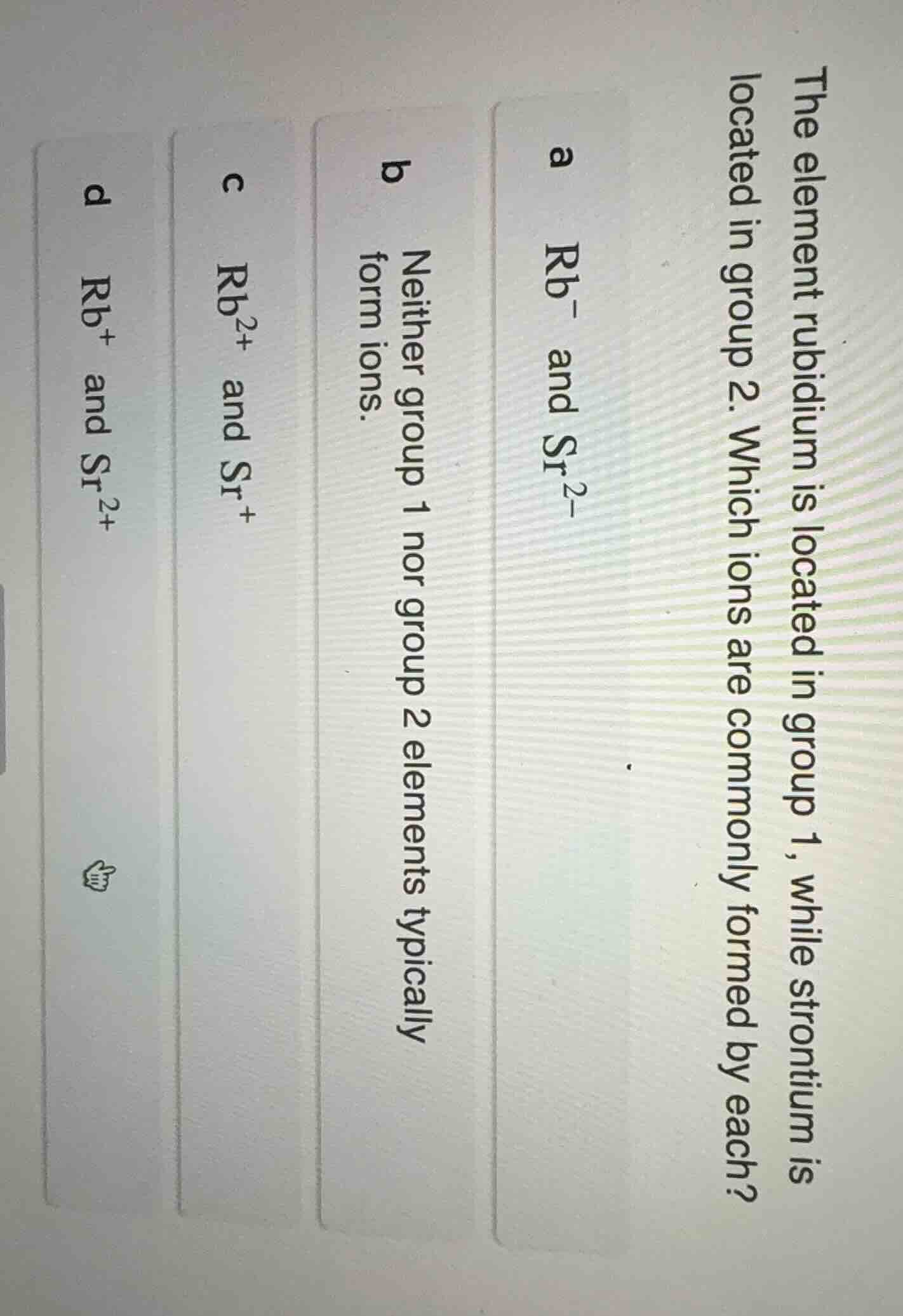
the element rubidium is located in group 1, while strontium is located in group 2. which ions are commonly formed by each?
a rb⁻ and sr²⁻
b neither group 1 nor group 2 elements typically form ions.
c rb²⁺ and sr⁺
d rb⁺ and sr²⁺
Brief Explanations
Group 1 elements (like Rb) have 1 valence electron, so they lose 1 electron to form +1 ions ($\text{Rb}^+$). Group 2 elements (like Sr) have 2 valence electrons, so they lose 2 electrons to form +2 ions ($\text{Sr}^{2+}$).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
d. $\text{Rb}^+$ and $\text{Sr}^{2+}$