QUESTION IMAGE
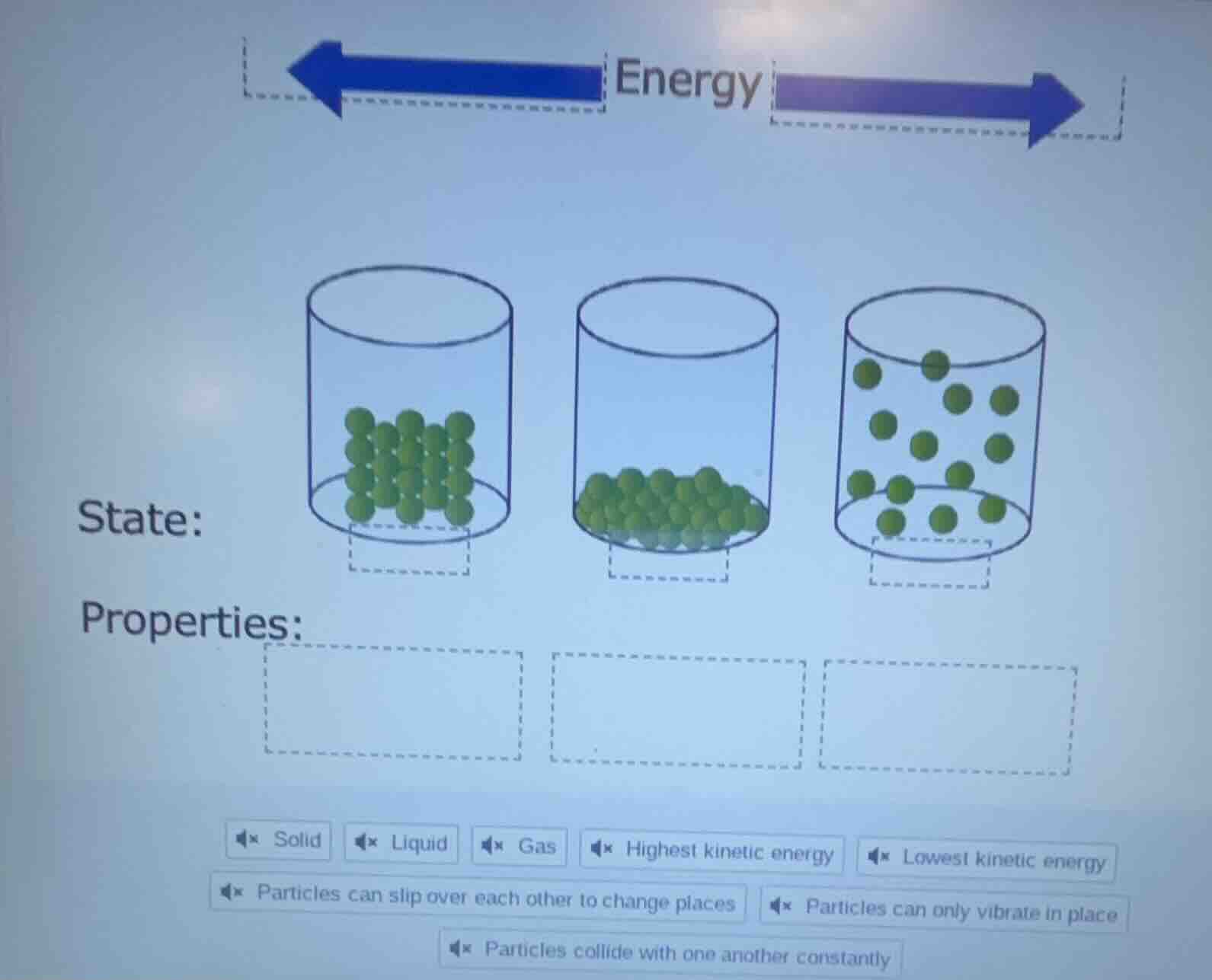
Question
energy
state:
properties:
solid
liquid
gas
highest kinetic energy
lowest kinetic energy
particles can slip over each other to change places
particles can only vibrate in place
particles collide with one another constantly
Brief Explanations
- First cylinder (tightly packed particles): This matches the structure of a solid, where particles are fixed in a rigid arrangement, only vibrate in place, and have the lowest kinetic energy of the three states.
- Second cylinder (loosely packed, flowing particles): This matches the structure of a liquid, where particles are close together but can slip past one another, allowing the substance to take the shape of its container.
- Third cylinder (spread out, free-moving particles): This matches the structure of a gas, where particles are far apart, move freely, constantly collide, and have the highest kinetic energy of the three states.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- First cylinder (left):
State: Solid
Properties: Lowest kinetic energy, Particles can only vibrate in place
- Second cylinder (middle):
State: Liquid
Properties: Particles can slip over each other to change places
- Third cylinder (right):
State: Gas
Properties: Highest kinetic energy, Particles collide with one another constantly