QUESTION IMAGE
Question
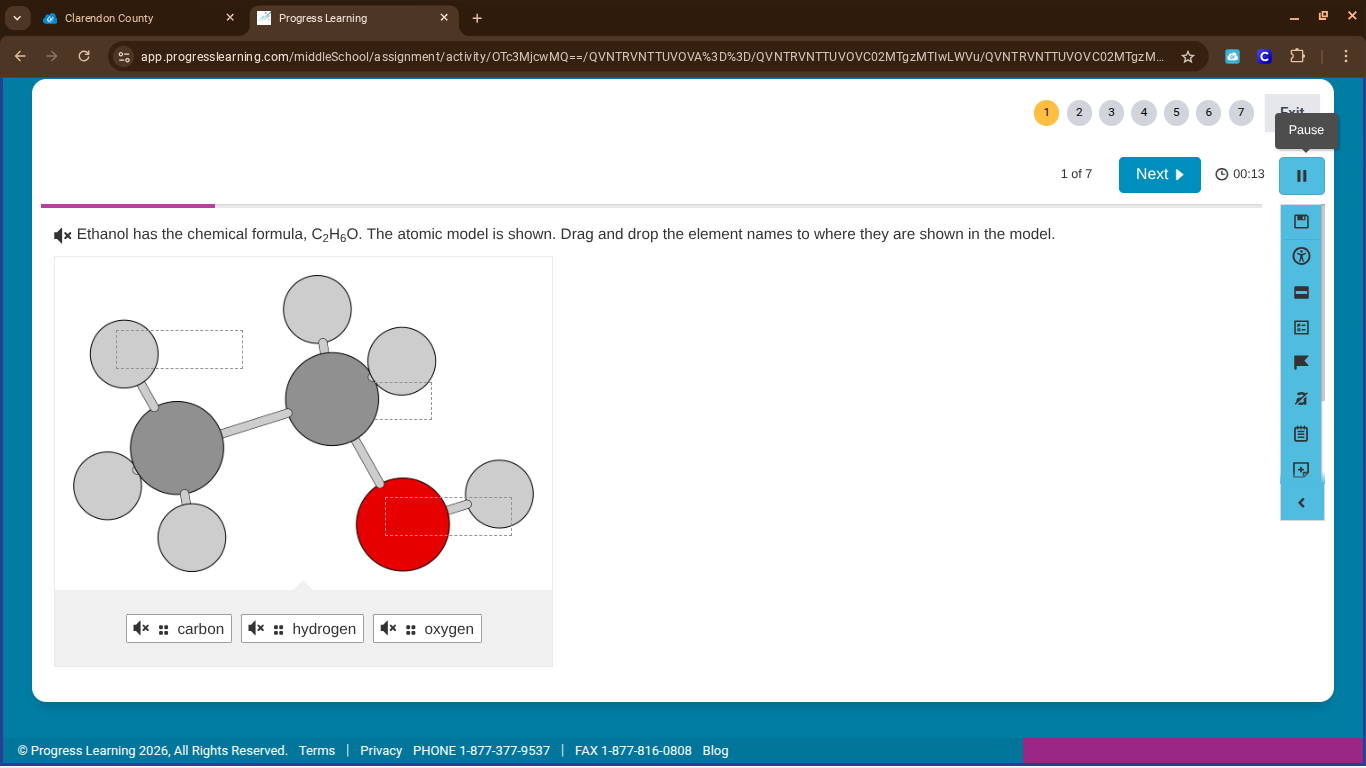
ethanol has the chemical formula, c₂h₆o. the atomic model is shown. drag and drop the element names to where they are shown in the model.
To solve this, we analyze the chemical formula of ethanol ($\ce{C_2H_6O}$) and the atomic model. Carbon (C) atoms are the larger gray ones (there are 2), hydrogen (H) atoms are the smaller gray ones (there are 6), and oxygen (O) is the red atom (1). So we match: the two larger gray atoms → carbon; the six smaller gray atoms → hydrogen; the red atom → oxygen. Then drag and drop each element name to its corresponding atom in the model.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Larger gray atoms: carbon
- Smaller gray atoms: hydrogen
- Red atom: oxygen
(Perform the drag - and - drop action: drag "carbon" to the larger gray atoms, "hydrogen" to the smaller gray atoms, and "oxygen" to the red atom in the atomic model of ethanol.)