QUESTION IMAGE
Question
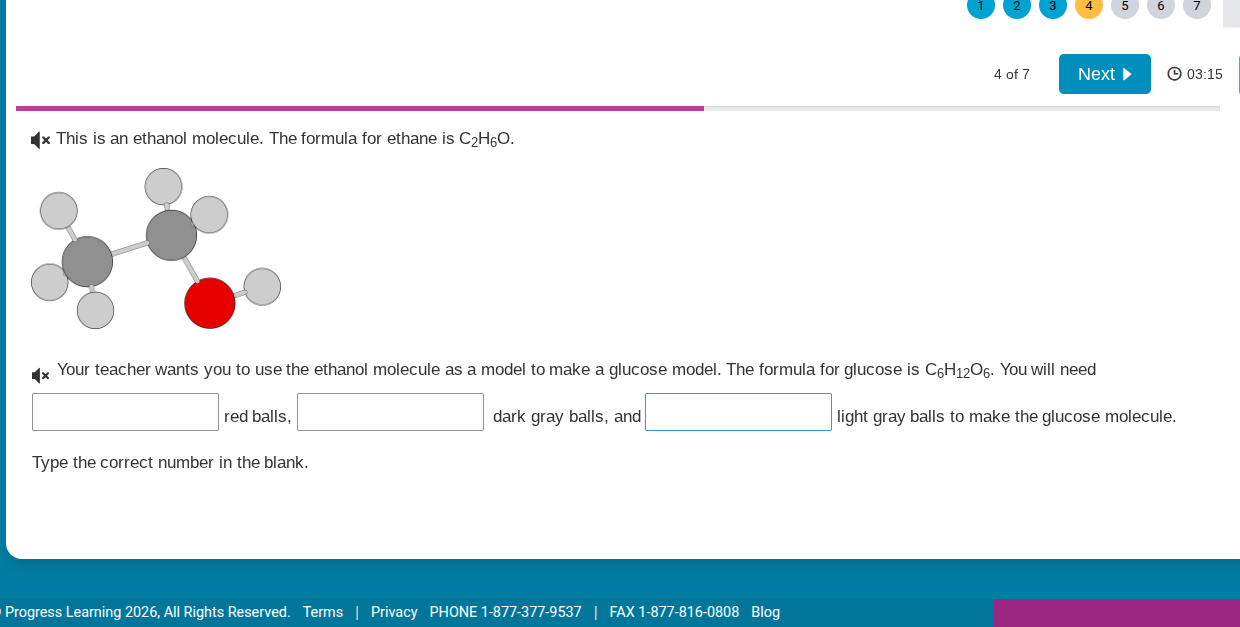
this is an ethanol molecule. the formula for ethane is $\ce{c_{2}h_{6}o}$. your teacher wants you to use the ethanol molecule as a model to make a glucose model. the formula for glucose is $\ce{c_{6}h_{12}o_{6}}$. you will need \boxed{} red balls, \boxed{} dark gray balls, and \boxed{} light gray balls to make the glucose molecule. type the correct number in the blank.
Step1: Match balls to elements
From the ethanol model: red = O, dark gray = C, light gray = H.
Step2: Extract counts from glucose formula
Glucose formula: $\text{C}_6\text{H}_{12}\text{O}_6$.
- O (red) count: $6$
- C (dark gray) count: $6$
- H (light gray) count: $12$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
6 red balls, 6 dark gray balls, and 12 light gray balls