QUESTION IMAGE
Question
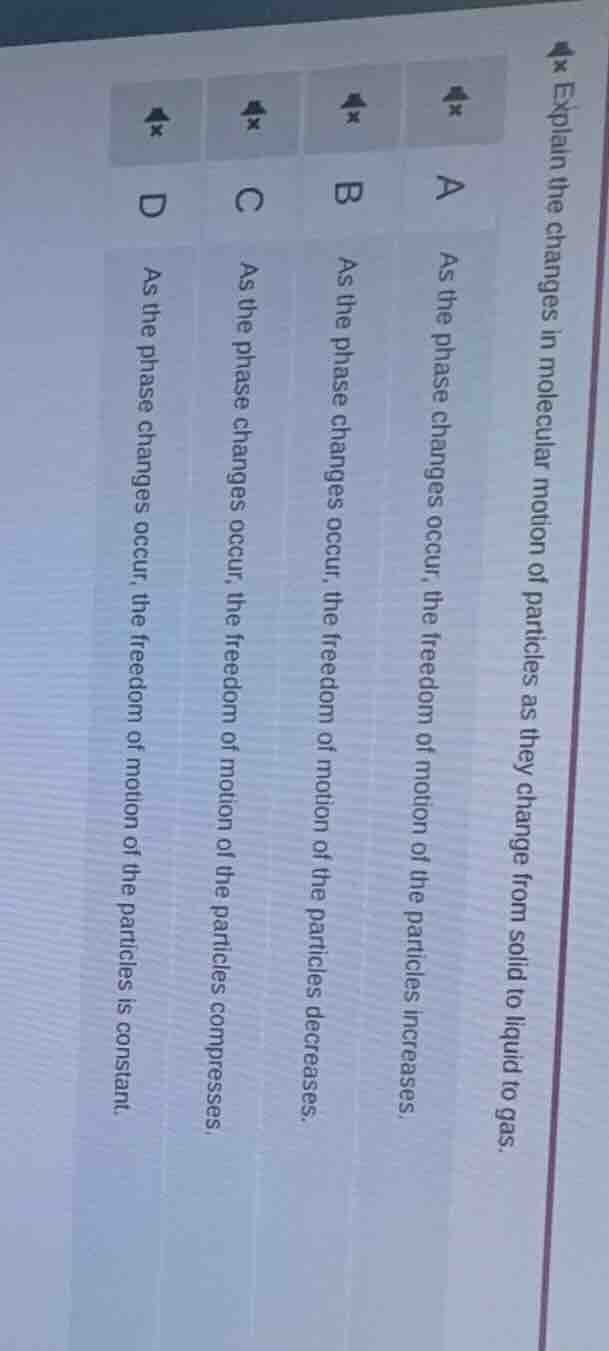
explain the changes in molecular motion of particles as they change from solid to liquid to gas. a as the phase changes occur, the freedom of motion of the particles increases. b as the phase changes occur, the freedom of motion of the particles decreases. c as the phase changes occur, the freedom of motion of the particles compresses. d as the phase changes occur, the freedom of motion of the particles is constant.
To determine the correct option, we analyze the molecular motion during phase changes:
- In a solid, particles are closely packed and have limited motion (vibrational).
- In a liquid, particles are more free to move (translational and rotational, with more space between them).
- In a gas, particles move freely and rapidly, with large spaces between them.
As a substance changes from solid to liquid to gas, the freedom of motion of the particles increases because the intermolecular forces weaken, and the space between particles increases, allowing for greater movement.
- Option A states that the freedom of motion increases, which matches our analysis.
- Option B says it decreases, which is incorrect (motion is more restricted in solids than liquids/gases).
- Option C says the motion "compresses," which is not a correct description of particle motion in phase changes.
- Option D says the freedom of motion is constant, which is incorrect as phase changes involve changes in particle arrangement and motion.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. As the phase changes occur, the freedom of motion of the particles increases.