QUESTION IMAGE
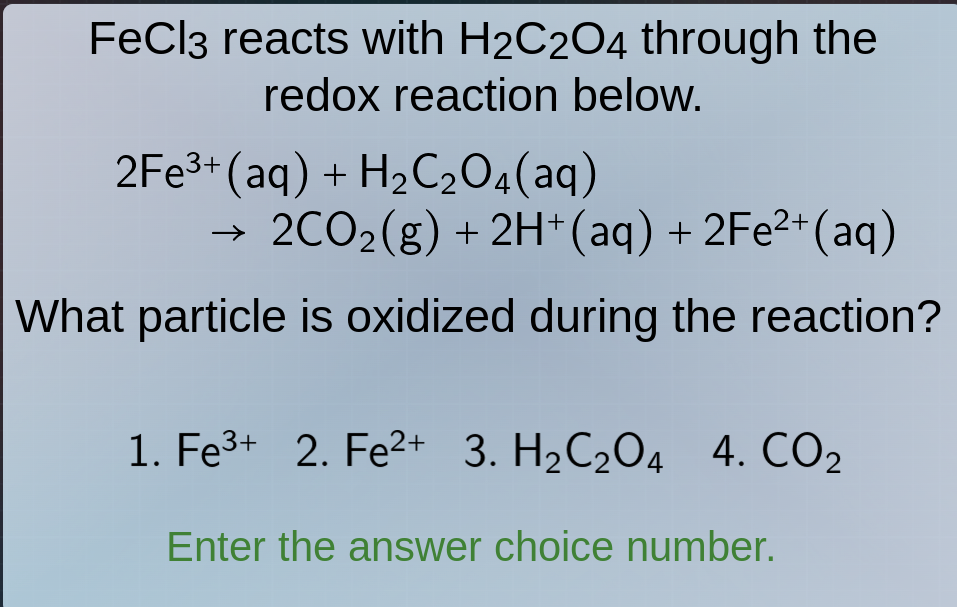
Question
fecl₃ reacts with h₂c₂o₄ through the redox reaction below.
2fe³⁺(aq) + h₂c₂o₄(aq) → 2co₂(g) + 2h⁺(aq) + 2fe²⁺(aq)
what particle is oxidized during the reaction?
- fe³⁺ 2. fe²⁺ 3. h₂c₂o₄ 4. co₂
enter the answer choice number.
To determine the oxidized particle, we analyze oxidation states. In \( \ce{H2C2O4} \), C has an oxidation state of +3. In \( \ce{CO2} \), C has an oxidation state of +4. An increase in oxidation state means oxidation. \( \ce{H2C2O4} \) (specifically C in it) has its oxidation state increase, so \( \ce{H2C2O4} \) is oxidized. \( \ce{Fe^{3+}} \) is reduced (oxidation state decreases from +3 to +2 in \( \ce{Fe^{2+}} \)), \( \ce{Fe^{2+}} \) is a product, and \( \ce{CO2} \) is an oxidation product, not the oxidized particle itself.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
3