QUESTION IMAGE
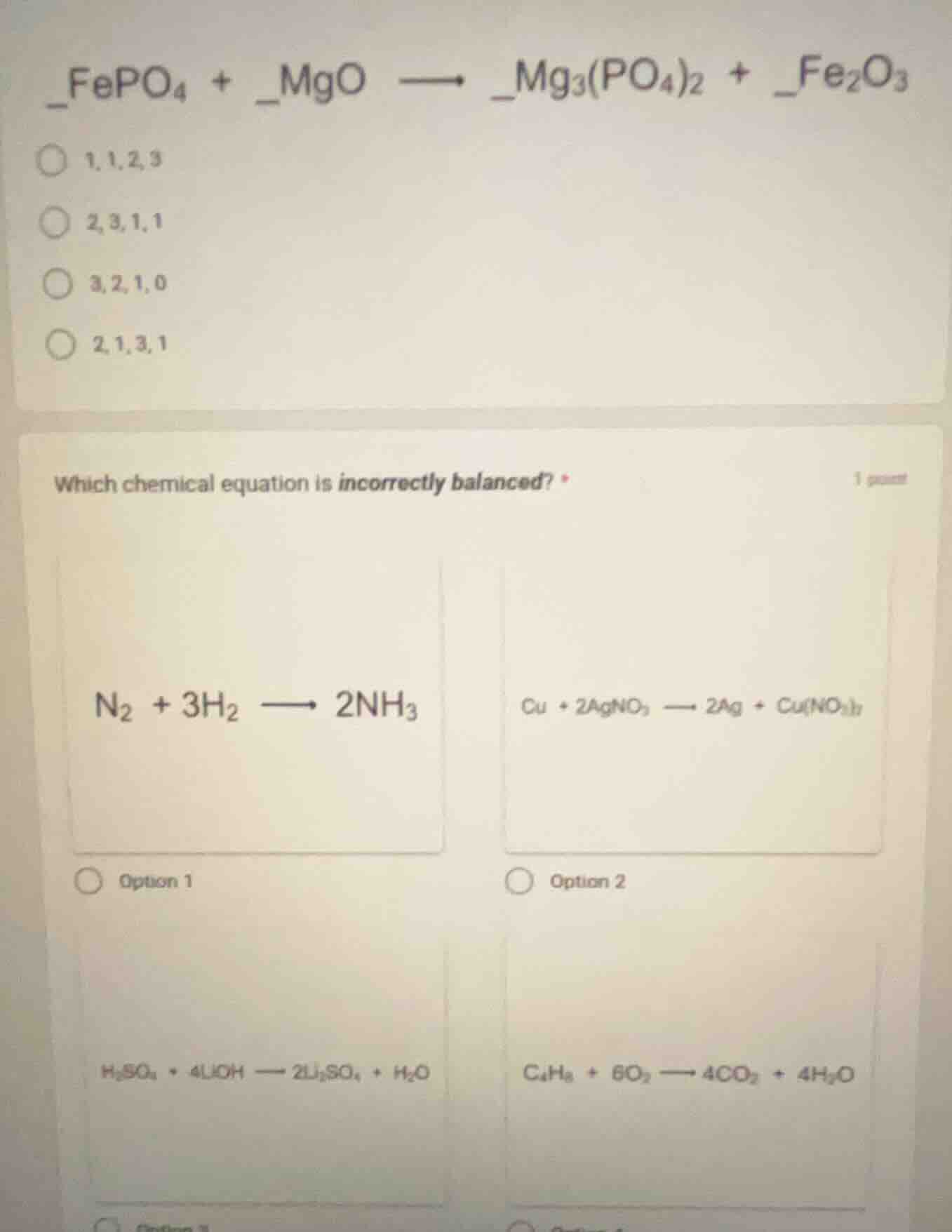
Question
_fepo₄ + _mgo → _mg₃(po₄)₂ + _fe₂o₃
○ 1,1,2,3
○ 2,3,1,1
○ 3,2,1,0
○ 2,1,3,1
which chemical equation is incorrectly balanced? *
n₂ + 3h₂ → 2nh₃ cu + 2agno₃ → 2ag + cu(no₃)₂
○ option 1 ○ option 2
h₂so₄ + 4lioh → 2li₂so₄ + h₂o c₄h₈ + 6o₂ → 4co₂ + 4h₂o
First Question (Balancing the Chemical Equation)
Step1: Balance phosphate ($\text{PO}_4^{3-}$) ions
There are 2 $\text{PO}_4^{3-}$ on the right, so add coefficient 2 to $\text{FePO}_4$:
$2\text{FePO}_4 + \text{MgO}
ightarrow \text{Mg}_3(\text{PO}_4)_2 + \text{Fe}_2\text{O}_3$
Step2: Balance Fe atoms
2 Fe on left matches 2 Fe on right, no change needed.
Step3: Balance Mg atoms
3 Mg on right, add coefficient 3 to $\text{MgO}$:
$2\text{FePO}_4 + 3\text{MgO}
ightarrow \text{Mg}_3(\text{PO}_4)_2 + \text{Fe}_2\text{O}_3$
Step4: Verify O atoms
Left: $(2\times4)+(3\times1)=11$; Right: $(2\times4)+(3\times1)=11$. Balanced.
- Option 1: $\text{N}_2 + 3\text{H}_2
ightarrow 2\text{NH}_3$: 2 N, 6 H on both sides; balanced.
- Option 2: $\text{Cu} + 2\text{AgNO}_3
ightarrow 2\text{Ag} + \text{Cu(NO}_3\text{)}_2$: 1 Cu, 2 Ag, 2 $\text{NO}_3^-$ on both sides; balanced.
- Option 3: $\text{H}_2\text{SO}_4 + 4\text{LiOH}
ightarrow 2\text{Li}_2\text{SO}_4 + \text{H}_2\text{O}$: Left has 1 $\text{SO}_4^{2-}$, right has 2 $\text{SO}_4^{2-}$; left has 4 Li, right has 4 Li, but H and O are unbalanced (left: 6 H, 8 O; right: 2 H, 9 O). This equation is incorrectly balanced.
- Option 4: $\text{C}_4\text{H}_8 + 6\text{O}_2
ightarrow 4\text{CO}_2 + 4\text{H}_2\text{O}$: 4 C, 8 H, 12 O on both sides; balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2, 3, 1, 1
---