QUESTION IMAGE
Question
- fill in the blank 1 point
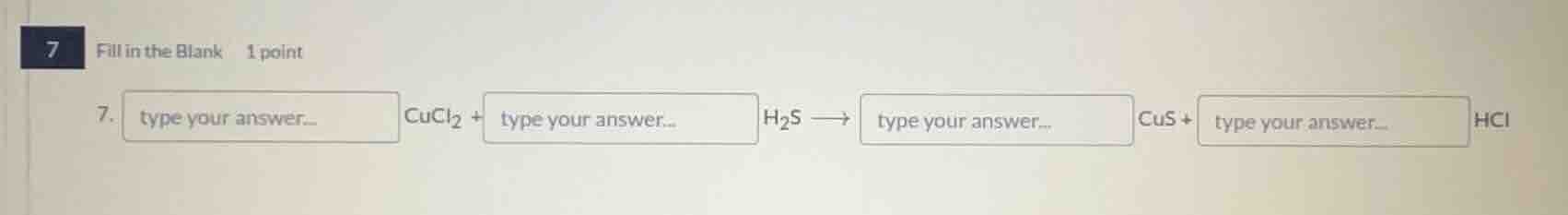
- type your answer... cucl₂ + type your answer... h₂s ⟶ type your answer... cus + type your answer... hcl
Step1: Identify the reaction type
This is a double - displacement reaction. In a double - displacement reaction, the general form is \(AB + CD
ightarrow AD+CB\). For the reaction between \(CuCl_2\) and \(H_2S\), we need to balance the equation.
First, let's write the unbalanced equation: \(CuCl_2 + H_2S
ightarrow CuS + HCl\)
Step2: Balance the chlorine and hydrogen atoms
On the left - hand side, we have 2 chlorine atoms in \(CuCl_2\) and 2 hydrogen atoms in \(H_2S\). On the right - hand side, in \(HCl\), if we put a coefficient of 2 in front of \(HCl\), the number of hydrogen and chlorine atoms will be balanced.
The balanced equation is: \(1CuCl_2+1H_2S
ightarrow 1CuS + 2HCl\)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1, 1, 1, 2 (The coefficients in order for \(CuCl_2\), \(H_2S\), \(CuS\), \(HCl\))