QUESTION IMAGE
Question
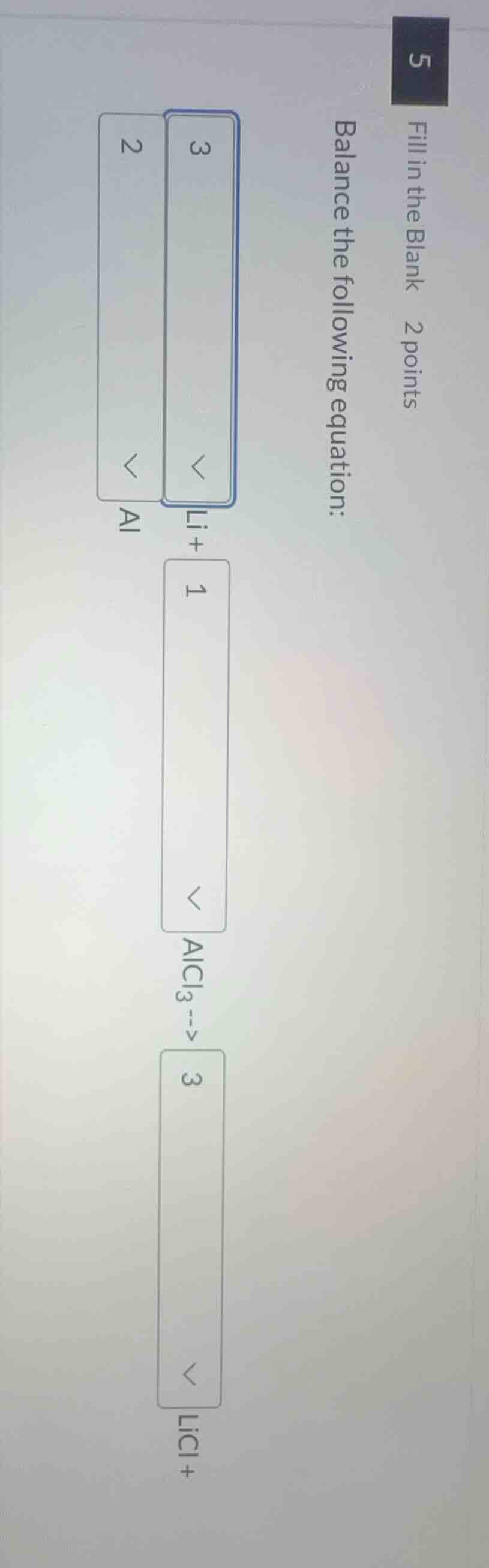
fill in the blank 2 points
balance the following equation:
3 li + 1 alcl₃ --> 3 licl + 2 al
Step1: Identify the reaction type
This is a single - displacement reaction where Li displaces Al from \(AlCl_3\). The unbalanced equation is \(Li + AlCl_3
ightarrow LiCl+Al\).
Step2: Balance Cl atoms
In \(AlCl_3\), there are 3 Cl atoms. In \(LiCl\), there is 1 Cl atom. To balance Cl, we put a coefficient of 3 in front of \(LiCl\). So the equation becomes \(Li + AlCl_3
ightarrow 3LiCl+Al\).
Step3: Balance Li atoms
Now, on the right - hand side, we have 3 Li atoms from \(3LiCl\). So we put a coefficient of 3 in front of Li on the left - hand side. The equation is \(3Li + AlCl_3
ightarrow 3LiCl+Al\).
Step4: Balance Al atoms
The number of Al atoms is 1 on both sides, so Al is already balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The balanced equation is \(3Li + 1AlCl_3
ightarrow 3LiCl + 1Al\) (or in the format of the blanks: for \(Li\): 3, for \(AlCl_3\): 1, for \(LiCl\): 3, for \(Al\): 1). But looking at the given blanks, the blank for \(Li\) is 3, for \(AlCl_3\) is 1, for \(LiCl\) is 3, and for \(Al\) is 1. If we consider the blanks in the order of the equation (assuming the equation is \( \text{[Li coefficient]}Li+\text{[AlCl}_3\text{ coefficient]}AlCl_3
ightarrow\text{[LiCl coefficient]}LiCl + \text{[Al coefficient]}Al\)), then the coefficients are 3, 1, 3, 1. But from the given options in the blanks (the first blank for Li is 3, then for \(AlCl_3\) the blank is 1, then for LiCl the blank is 3, and for Al the blank is 1). However, if we look at the Al blank (the second blank in the left - hand side, the one with 2 as an option, but our calculation shows 1 for Al? Wait, no, the Al on the left - hand side? Wait, no, the original equation is \(Li + AlCl_3
ightarrow LiCl + Al\). The Al is on the product side. Wait, maybe the equation is written as \(Li+AlCl_3
ightarrow LiCl + Al\), and we need to fill the coefficients. So the correct coefficients are: for Li: 3, for \(AlCl_3\): 1, for \(LiCl\): 3, for Al: 1. But in the given blanks, the first blank (Li) is 3, the second blank (AlCl_3) is 1, the third blank (LiCl) is 3, and the fourth blank (Al) is 1. If we consider the Al blank (the one with 2 as an option, but our balance shows 1 for Al). Wait, maybe there is a mis - writing in the problem's blank arrangement. But based on the balancing, the coefficient for \(Li\) is 3, for \(AlCl_3\) is 1, for \(LiCl\) is 3, and for \(Al\) is 1.