QUESTION IMAGE
Question
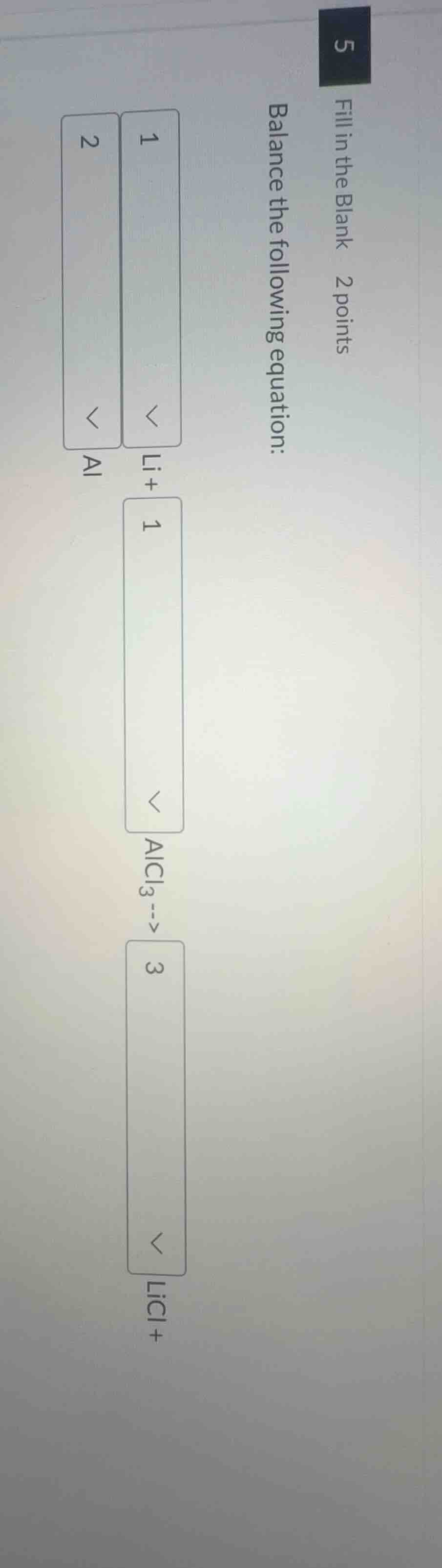
fill in the blank 2 points
balance the following equation:
1
li+ 1
alcl₃--> 3
licl+ 2
al
Step1: Identify atoms to balance
We have the equation (unbalanced): $ \text{Li} + \text{AlCl}_3
ightarrow \text{LiCl} + \text{Al} $
First, look at Cl atoms. On the left, in $\text{AlCl}_3$, there are 3 Cl. On the right, in $\text{LiCl}$, there is 1 Cl. So we need to put a coefficient of 3 in front of $\text{LiCl}$ to balance Cl: $ \text{Li} + \text{AlCl}_3
ightarrow 3\text{LiCl} + \text{Al} $
Step2: Balance Li atoms
Now, Li on the right is 3 (from $3\text{LiCl}$), so we put 3 in front of Li on the left: $ 3\text{Li} + \text{AlCl}_3
ightarrow 3\text{LiCl} + \text{Al} $
Step3: Balance Al atoms
Al on the left is 1 (from $\text{AlCl}_3$) and on the right is 1 (from $\text{Al}$), so Al is already balanced.
Wait, but the problem's initial blanks: Let's re - express the equation with the given blanks. The equation structure is: $ \underline{1}\text{Li} + \underline{1}\text{AlCl}_3
ightarrow \underline{3}\text{LiCl} + \underline{1}\text{Al} $? No, wait the user's interface has blanks: first blank (Li), second (Al), third (LiCl). Wait, maybe the correct balanced equation is $ 3\text{Li} + \text{AlCl}_3 = 3\text{LiCl} + \text{Al} $. But the given blanks: first blank (Li) has a default 1, second (Al) default 2? Wait, maybe I misread. Let's check the atoms again.
Wait, the original equation in the problem (from the image) is: $ \underline{1}\text{Li} + \underline{2}\text{Al} + \underline{1}\text{AlCl}_3
ightarrow \underline{3}\text{LiCl} +...$ No, no, the correct approach is to balance the equation properly.
The correct balanced chemical equation for the reaction between lithium (Li) and aluminium chloride ($\text{AlCl}_3$) is a single - displacement reaction:
- Balance Cl: There are 3 Cl in $\text{AlCl}_3$, so we need 3 $\text{LiCl}$ (since each $\text{LiCl}$ has 1 Cl). So put 3 in front of $\text{LiCl}$.
- Balance Li: Since we have 3 $\text{LiCl}$, we need 3 Li on the left, so put 3 in front of Li.
- Balance Al: There is 1 Al in $\text{AlCl}_3$ and 1 Al on the right, so Al is balanced.
So the balanced equation is $ 3\text{Li} + \text{AlCl}_3 = 3\text{LiCl} + \text{Al} $
But the given blanks:
- For Li: the first blank (Li) should be 3, but it's given as 1? Wait, maybe the problem's blanks are for the coefficients in the order: Li, Al, $\text{AlCl}_3$, $\text{LiCl}$, Al. Wait, no, the image shows:
First blank: Li (with 1 as default)
Second blank: Al (with 2 as default)
Third blank: $\text{AlCl}_3$ (with 1 as default)
Fourth blank: $\text{LiCl}$ (with 3 as default)
Fifth blank: Al (?)
Wait, maybe the correct coefficients are:
Li: 3, Al: 1, $\text{AlCl}_3$: 1, $\text{LiCl}$: 3, Al: 1.
But the user's blanks:
First blank (Li): should be 3 (but given 1, maybe a mistake, but following the balancing steps, the coefficient for Li is 3, for Al is 1, for $\text{LiCl}$ is 3.
But the problem's interface has:
1 (Li) + 2 (Al) + 1 ($\text{AlCl}_3$) -> 3 ($\text{LiCl}$) +...
Wait, no, the correct balanced equation is $ 3\text{Li} + \text{AlCl}_3 = 3\text{LiCl} + \text{Al} $. So the coefficients are: Li: 3, $\text{AlCl}_3$: 1, $\text{LiCl}$: 3, Al: 1.
If we consider the blanks:
- Coefficient of Li: 3
- Coefficient of Al: 1
- Coefficient of $\text{LiCl}$: 3
But the given blanks: first (Li) has 1, second (Al) has 2, third ($\text{LiCl}$) has 3. Maybe there was a mis - formatting. But following the balancing rules:
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The balanced equation is $ \boldsymbol{3}\text{Li} + \text{AlCl}_3 = 3\text{LiCl} + \text{Al} $, so the coefficients (for the blanks in order: Li, Al, LiCl) should be 3, 1, 3. But if we follow the given blank structure (with first Li as 1, second Al as 2, third LiCl as 3), there might be a mistake, but the correct balanced coefficients are 3 (Li), 1 (Al), 3 (LiCl).