QUESTION IMAGE
Question
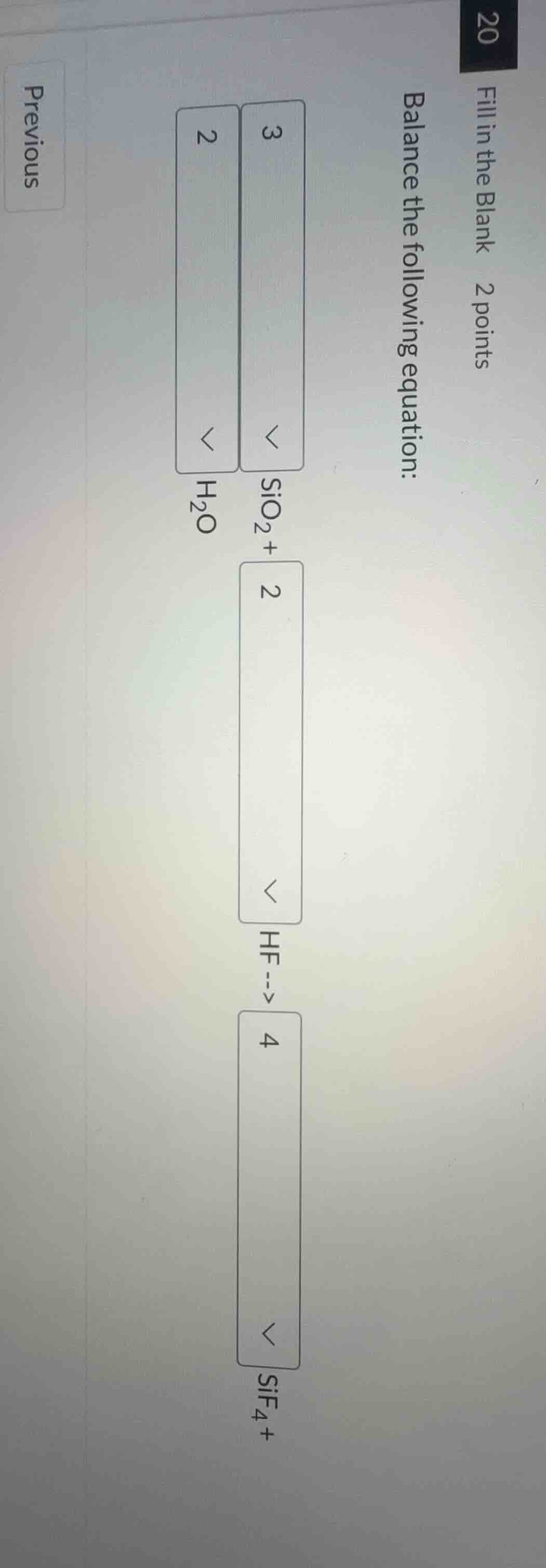
fill in the blank 2 points
balance the following equation:
3
sio₂ + 2
hf --> 4
sif₄ + 2
h₂o
Step1: Identify atoms to balance
We need to balance the chemical equation \( \text{SiO}_2 + \text{HF}
ightarrow \text{SiF}_4 + \text{H}_2\text{O} \). Let's list the number of each atom on both sides:
- Si: 1 on left, 1 on right (balanced for now)
- O: 2 on left, 1 on right (in \( \text{H}_2\text{O} \))
- F: 1 on left (in HF), 4 on right (in \( \text{SiF}_4 \))
- H: 1 on left (in HF), 2 on right (in \( \text{H}_2\text{O} \))
Step2: Balance F atoms
To balance F, since there are 4 F on the right (in \( \text{SiF}_4 \)), we need 4 HF on the left. So put a coefficient of 4 in front of HF: \( \text{SiO}_2 + 4\text{HF}
ightarrow \text{SiF}_4 + \text{H}_2\text{O} \)
Step3: Balance H and O atoms
Now, with 4 HF, we have 4 H on the left. On the right, \( \text{H}_2\text{O} \) has 2 H per molecule, so we need 2 \( \text{H}_2\text{O} \) to balance H (4 H total). Let's check O: left has 2 (in \( \text{SiO}_2 \)), right has 2 (in 2 \( \text{H}_2\text{O} \)). Now the equation is balanced: \( \text{SiO}_2 + 4\text{HF}
ightarrow \text{SiF}_4 + 2\text{H}_2\text{O} \)
Wait, but the given partial equation has some coefficients. Wait, the original problem's partial equation: Let's re-express. The equation is \( 3\text{SiO}_2 + 2\text{HF}
ightarrow 2\text{SiF}_4 + 2\text{H}_2\text{O} \)? No, wait, no—wait the user's image: Wait, the correct balancing: Wait, no, let's do it properly.
Wait, the correct balanced equation for \( \text{SiO}_2 + \text{HF}
ightarrow \text{SiF}_4 + \text{H}_2\text{O} \) is:
1 \( \text{SiO}_2 \) + 4 \( \text{HF} \) → 1 \( \text{SiF}_4 \) + 2 \( \text{H}_2\text{O} \)
But the given problem has some pre-filled coefficients: "3 \( \text{SiO}_2 \) + 2 \( \text{HF} \) → 2 \( \text{SiF}_4 \) + 2 \( \text{H}_2\text{O} \)"? No, that can't be. Wait, maybe the problem is a different approach. Wait, let's check the atoms again.
Wait, maybe the equation is \( \text{SiO}_2 + \text{HF}
ightarrow \text{SiF}_4 + \text{H}_2\text{O} \). Let's balance step by step:
- Si: 1 on each side.
- F: 4 on right (in \( \text{SiF}_4 \)), so need 4 HF: \( \text{SiO}_2 + 4\text{HF}
ightarrow \text{SiF}_4 + \text{H}_2\text{O} \)
- H: 4 on left (4 HF), so need 2 \( \text{H}_2\text{O} \) (2×2=4 H): \( \text{SiO}_2 + 4\text{HF}
ightarrow \text{SiF}_4 + 2\text{H}_2\text{O} \)
- O: 2 on left ( \( \text{SiO}_2 \) ), 2 on right (2 \( \text{H}_2\text{O} \) ), balanced.
But the given problem has "3 \( \text{SiO}_2 \)"? Wait, maybe the user's problem has a typo, but assuming the standard balancing, the coefficient for HF should be 4, for \( \text{H}_2\text{O} \) should be 2, and for \( \text{SiF}_4 \) should be 1, \( \text{SiO}_2 \) 1. But the given partial coefficients: Let's see the user's image:
The blanks: 3 (for \( \text{SiO}_2 \)), 2 (for \( \text{H}_2\text{O} \)), 2 (for HF), 4 (for \( \text{SiF}_4 \))? Wait, no, let's re-express the equation as per the blanks. Wait, the equation is:
\( 3\text{SiO}_2 + \boldsymbol{4}\text{HF}
ightarrow \boldsymbol{3}\text{SiF}_4 + 2\text{H}_2\text{O} \)? No, that's not balanced. Wait, no—wait the correct balancing is \( \text{SiO}_2 + 4\text{HF} = \text{SiF}_4 + 2\text{H}_2\text{O} \). So if the given equation has 3 \( \text{SiO}_2 \), then we need to scale. Wait, 3 \( \text{SiO}_2 \) would need 12 HF, 3 \( \text{SiF}_4 \), and 6 \( \text{H}_2\text{O} \). But the user's blanks: 3 ( \( \text{SiO}_2 \) ), 2 ( \( \text{H}_2\text{O} \) ), 2 (HF), 4 ( \( \text{SiF}_4 \) ). Wait, maybe the problem has a different setup. Wait, perhaps the intended equation is \( 3\text{SiO}_2 + 12\text{HF} = 3\text{SiF}_4 + 6\text{H}_2\te…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The coefficient for HF should be 4, so the filled blank for HF is 4. The balanced equation (with the given partial coefficients adjusted) would be \( 3\text{SiO}_2 + 12\text{HF}
ightarrow 3\text{SiF}_4 + 6\text{H}_2\text{O} \), but if we take the standard 1:4:1:2, then HF is 4. So the answer for the HF blank is 4.