QUESTION IMAGE
Question
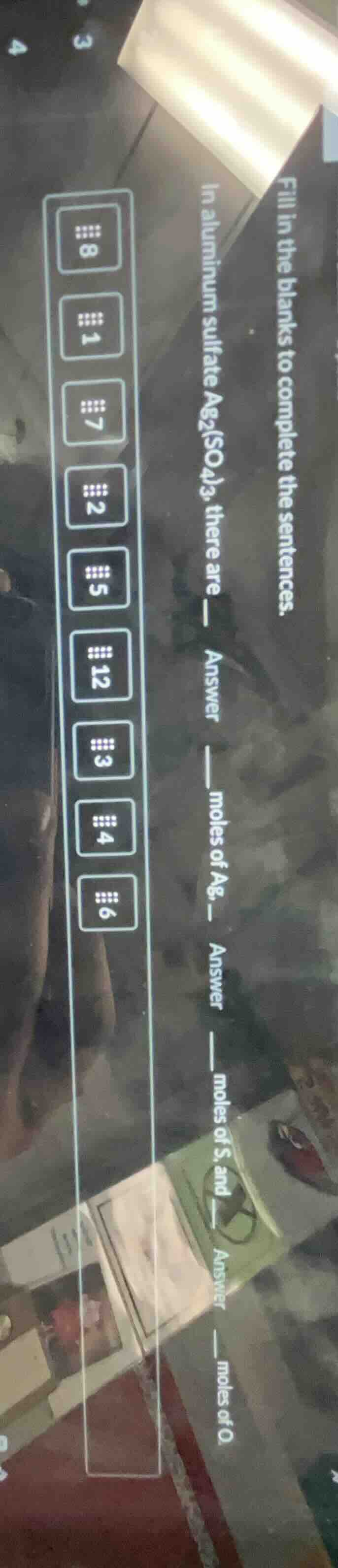
fill in the blanks to complete the sentences.
in aluminum sulfate $\text{al}_2(\text{so}_4)_3$, there are answer moles of ag, answer moles of s, and answer moles of o.
number options: 8, 1, 7, 2, 5, 12, 3, 4, 6
Step1: Identify Ag mole count
The subscript of Ag in $\text{Ag}_2(\text{SO}_4)_3$ is 2, so moles of Ag = 2.
Step2: Identify S mole count
Inside the parentheses, S has a subscript of 1, multiplied by the outer subscript 3: $1 \times 3 = 3$, so moles of S = 3.
Step3: Identify O mole count
Inside the parentheses, O has a subscript of 4, multiplied by the outer subscript 3: $4 \times 3 = 12$, so moles of O = 12.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2 moles of Ag, 3 moles of S, and 12 moles of O