QUESTION IMAGE
Question
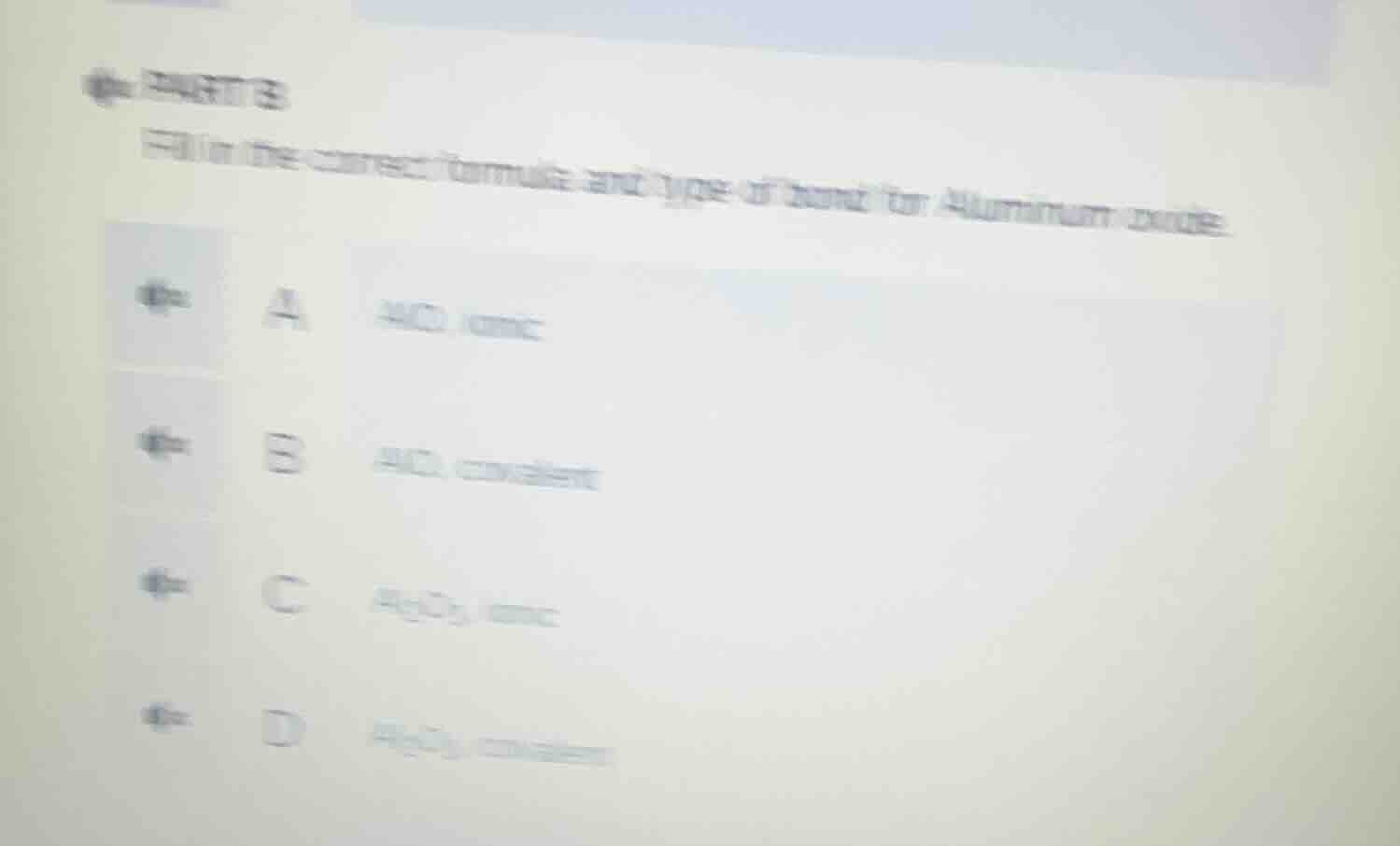
fill in the correct formula and type of bond for aluminium oxide. a alo ionic b alo covalent c al₂o₃ ionic d al₂o₃ covalent
Step1: Determine ion charges
Aluminum forms $\text{Al}^{3+}$, oxygen forms $\text{O}^{2-}$.
Step2: Balance charges for formula
Cross charges: $2\times\text{Al}^{3+}$ and $3\times\text{O}^{2-}$ give $\text{Al}_2\text{O}_3$.
Step3: Identify bond type
Bond between metal (Al) and nonmetal (O) is ionic.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. $\text{Al}_2\text{O}_3$, ionic