QUESTION IMAGE
Question
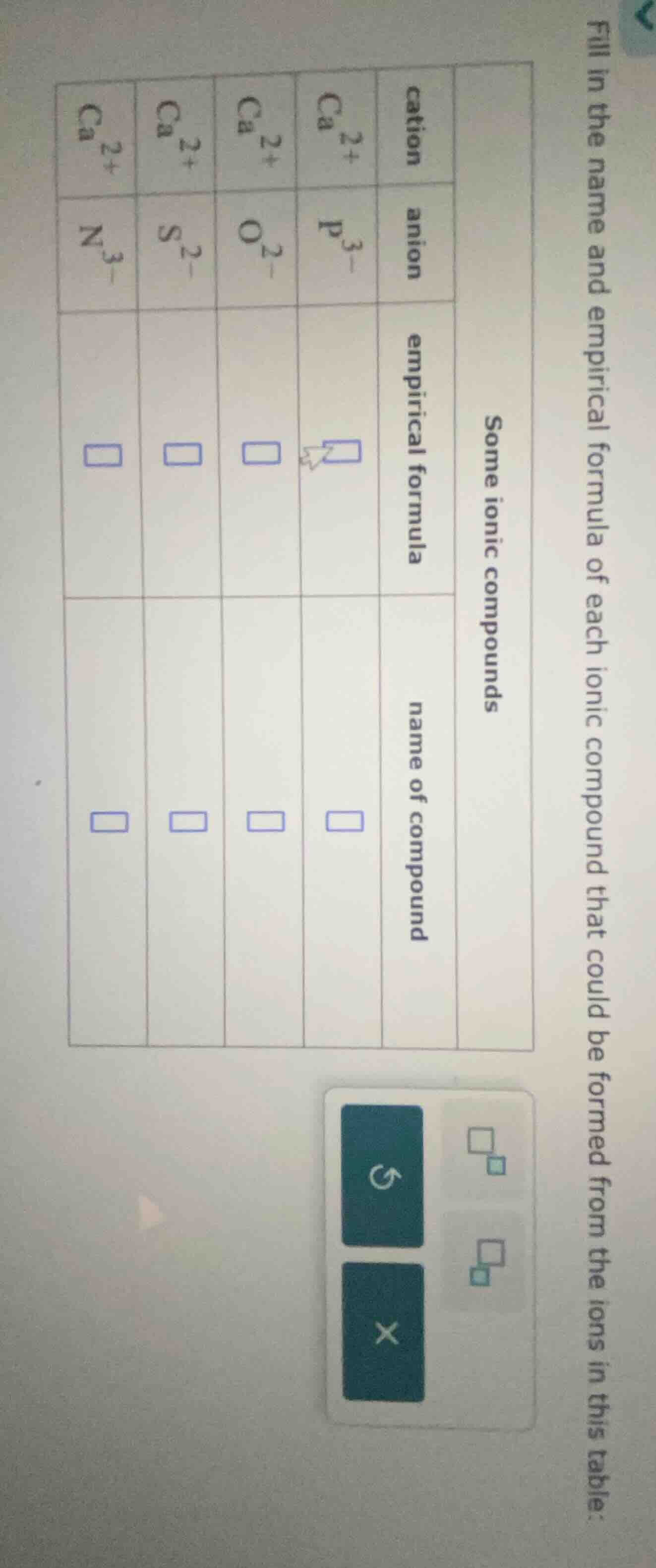
fill in the name and empirical formula of each ionic compound that could be formed from the ions in this table:
some ionic compounds
cation \tanion \tempirical formula \tname of compound
$ce{ca^{2+}}$ \t$ce{p^{3-}}$ \t$square$ \t$square$
$ce{ca^{2+}}$ \t$ce{o^{2-}}$ \t$square$ \t$square$
$ce{ca^{2+}}$ \t$ce{s^{2-}}$ \t$square$ \t$square$
$ce{ca^{2+}}$ \t$ce{n^{3-}}$ \t$square$ \t$square$
To solve for the empirical formula and name of each ionic compound, we use the criss - cross method (where the charge of the cation becomes the subscript of the anion and vice - versa, then simplify if needed) and the naming rules for ionic compounds (name the cation first, then the anion with an - ide suffix for monatomic anions).
For the first row (cation $\ce{Ca^{2+}}$, anion $\ce{P^{3-}}$)
Step 1: Determine the subscripts
The charge of $\ce{Ca^{2+}}$ is $2$ and the charge of $\ce{P^{3-}}$ is $3$. Using the criss - cross method, the subscript of $\ce{Ca}$ will be $3$ (the magnitude of the anion's charge) and the subscript of $\ce{P}$ will be $2$ (the magnitude of the cation's charge). So the empirical formula is $\ce{Ca_3P_2}$.
Step 2: Name the compound
The cation is calcium ($\ce{Ca^{2+}}$) and the anion is phosphide ($\ce{P^{3-}}$). So the name of the compound is calcium phosphide.
For the second row (cation $\ce{Ca^{2+}}$, anion $\ce{O^{2-}}$)
Step 1: Determine the subscripts
The charge of $\ce{Ca^{2+}}$ is $2$ and the charge of $\ce{O^{2-}}$ is $2$. When we apply the criss - cross method, we get $\ce{Ca_2O_2}$. But we can simplify this by dividing both subscripts by their greatest common divisor, which is $2$. So the empirical formula is $\ce{CaO}$.
Step 2: Name the compound
The cation is calcium ($\ce{Ca^{2+}}$) and the anion is oxide ($\ce{O^{2-}}$). So the name of the compound is calcium oxide.
For the third row (cation $\ce{Ca^{2+}}$, anion $\ce{S^{2-}}$)
Step 1: Determine the subscripts
The charge of $\ce{Ca^{2+}}$ is $2$ and the charge of $\ce{S^{2-}}$ is $2$. Using the criss - cross method, we initially get $\ce{Ca_2S_2}$. Dividing both subscripts by $2$ (their greatest common divisor), the empirical formula is $\ce{CaS}$.
Step 2: Name the compound
The cation is calcium ($\ce{Ca^{2+}}$) and the anion is sulfide ($\ce{S^{2-}}$). So the name of the compound is calcium sulfide.
For the fourth row (cation $\ce{Ca^{2+}}$, anion $\ce{N^{3-}}$)
Step 1: Determine the subscripts
The charge of $\ce{Ca^{2+}}$ is $2$ and the charge of $\ce{N^{3-}}$ is $3$. Using the criss - cross method, the subscript of $\ce{Ca}$ is $3$ and the subscript of $\ce{N}$ is $2$. So the empirical formula is $\ce{Ca_3N_2}$.
Step 2: Name the compound
The cation is calcium ($\ce{Ca^{2+}}$) and the anion is nitride ($\ce{N^{3-}}$). So the name of the compound is calcium nitride.
Summary Table
| cation | anion | empirical formula | name of compound |
|---|---|---|---|
| $\ce{Ca^{2+}}$ | $\ce{O^{2-}}$ | $\ce{CaO}$ | calcium oxide |
| $\ce{Ca^{2+}}$ | $\ce{S^{2-}}$ | $\ce{CaS}$ | calcium sulfide |
| $\ce{Ca^{2+}}$ | $\ce{N^{3-}}$ | $\ce{Ca_3N_2}$ | calcium nitride |
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve for the empirical formula and name of each ionic compound, we use the criss - cross method (where the charge of the cation becomes the subscript of the anion and vice - versa, then simplify if needed) and the naming rules for ionic compounds (name the cation first, then the anion with an - ide suffix for monatomic anions).
For the first row (cation $\ce{Ca^{2+}}$, anion $\ce{P^{3-}}$)
Step 1: Determine the subscripts
The charge of $\ce{Ca^{2+}}$ is $2$ and the charge of $\ce{P^{3-}}$ is $3$. Using the criss - cross method, the subscript of $\ce{Ca}$ will be $3$ (the magnitude of the anion's charge) and the subscript of $\ce{P}$ will be $2$ (the magnitude of the cation's charge). So the empirical formula is $\ce{Ca_3P_2}$.
Step 2: Name the compound
The cation is calcium ($\ce{Ca^{2+}}$) and the anion is phosphide ($\ce{P^{3-}}$). So the name of the compound is calcium phosphide.
For the second row (cation $\ce{Ca^{2+}}$, anion $\ce{O^{2-}}$)
Step 1: Determine the subscripts
The charge of $\ce{Ca^{2+}}$ is $2$ and the charge of $\ce{O^{2-}}$ is $2$. When we apply the criss - cross method, we get $\ce{Ca_2O_2}$. But we can simplify this by dividing both subscripts by their greatest common divisor, which is $2$. So the empirical formula is $\ce{CaO}$.
Step 2: Name the compound
The cation is calcium ($\ce{Ca^{2+}}$) and the anion is oxide ($\ce{O^{2-}}$). So the name of the compound is calcium oxide.
For the third row (cation $\ce{Ca^{2+}}$, anion $\ce{S^{2-}}$)
Step 1: Determine the subscripts
The charge of $\ce{Ca^{2+}}$ is $2$ and the charge of $\ce{S^{2-}}$ is $2$. Using the criss - cross method, we initially get $\ce{Ca_2S_2}$. Dividing both subscripts by $2$ (their greatest common divisor), the empirical formula is $\ce{CaS}$.
Step 2: Name the compound
The cation is calcium ($\ce{Ca^{2+}}$) and the anion is sulfide ($\ce{S^{2-}}$). So the name of the compound is calcium sulfide.
For the fourth row (cation $\ce{Ca^{2+}}$, anion $\ce{N^{3-}}$)
Step 1: Determine the subscripts
The charge of $\ce{Ca^{2+}}$ is $2$ and the charge of $\ce{N^{3-}}$ is $3$. Using the criss - cross method, the subscript of $\ce{Ca}$ is $3$ and the subscript of $\ce{N}$ is $2$. So the empirical formula is $\ce{Ca_3N_2}$.
Step 2: Name the compound
The cation is calcium ($\ce{Ca^{2+}}$) and the anion is nitride ($\ce{N^{3-}}$). So the name of the compound is calcium nitride.
Summary Table
| cation | anion | empirical formula | name of compound |
|---|---|---|---|
| $\ce{Ca^{2+}}$ | $\ce{O^{2-}}$ | $\ce{CaO}$ | calcium oxide |
| $\ce{Ca^{2+}}$ | $\ce{S^{2-}}$ | $\ce{CaS}$ | calcium sulfide |
| $\ce{Ca^{2+}}$ | $\ce{N^{3-}}$ | $\ce{Ca_3N_2}$ | calcium nitride |