QUESTION IMAGE
Question
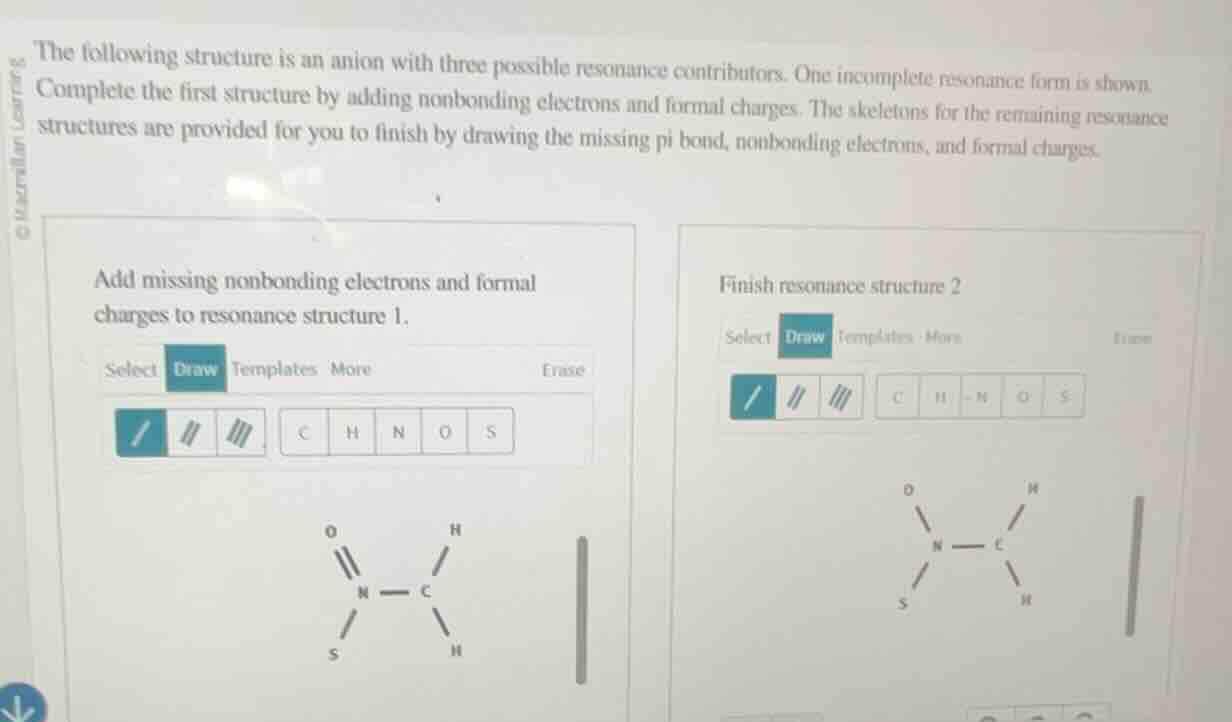
the following structure is an anion with three possible resonance contributors. one incomplete resonance form is shown. complete the first structure by adding nonbonding electrons and formal charges. the skeletons for the remaining resonance structures are provided for you to finish by drawing the missing pi bond, nonbonding electrons, and formal charges. add missing nonbonding electrons and formal charges to resonance structure 1. finish resonance structure 2
Step1: Calculate formal charge for N
Formal charge formula: $FC = V - N - \frac{B}{2}$, where $V$ = valence electrons, $N$ = nonbonding electrons, $B$ = bonding electrons.
For N (valence e⁻=5): Bonding e⁻=6 (double bond + single bond), add 2 nonbonding e⁻.
$FC = 5 - 2 - \frac{6}{2} = 0$
Step2: Add nonbonding e⁻ to O
O has double bond to N (4 bonding e⁻), valence e⁻=6. Add 4 nonbonding e⁻.
$FC = 6 - 4 - \frac{4}{2} = 0$
Step3: Calculate formal charge for S
S has single bond to N (2 bonding e⁻), valence e⁻=6. Add 6 nonbonding e⁻.
$FC = 6 - 6 - \frac{2}{2} = -1$
Step4: Resonance Structure 2: Add pi bond
Move pi bond from N=O to N-S, making N=S.
Step5: Add nonbonding e⁻ to Structure 2 O
O now has single bond to N (2 bonding e⁻), add 6 nonbonding e⁻.
$FC = 6 - 6 - \frac{2}{2} = -1$
Step6: Add nonbonding e⁻ to Structure 2 S
S has double bond to N (4 bonding e⁻), add 4 nonbonding e⁻.
$FC = 6 - 4 - \frac{4}{2} = 0$
Step7: Confirm N charge in Structure 2
N has single bond to O, double bond to S, single bond to C (6 bonding e⁻), 2 nonbonding e⁻.
$FC = 5 - 2 - \frac{6}{2} = 0$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Resonance Structure 1 (Completed):
- N: 2 nonbonding electrons (1 lone pair), formal charge = 0
- O: 4 nonbonding electrons (2 lone pairs), formal charge = 0
- S: 6 nonbonding electrons (3 lone pairs), formal charge = -1
- CH₃ group: No formal charges, all H have 0 nonbonding electrons
Resonance Structure 2 (Completed):
- Pi bond between N and S (N=S)
- O: 6 nonbonding electrons (3 lone pairs), formal charge = -1
- S: 4 nonbonding electrons (2 lone pairs), formal charge = 0
- N: 2 nonbonding electrons (1 lone pair), formal charge = 0
- CH₃ group: No formal charges, all H have 0 nonbonding electrons
(Third resonance contributor would have the pi bond between N-C, with S having 6 nonbonding e⁻ (FC=-1), O having 4 nonbonding e⁻ (FC=0), N having 2 nonbonding e⁻ (FC=0))