QUESTION IMAGE
Question
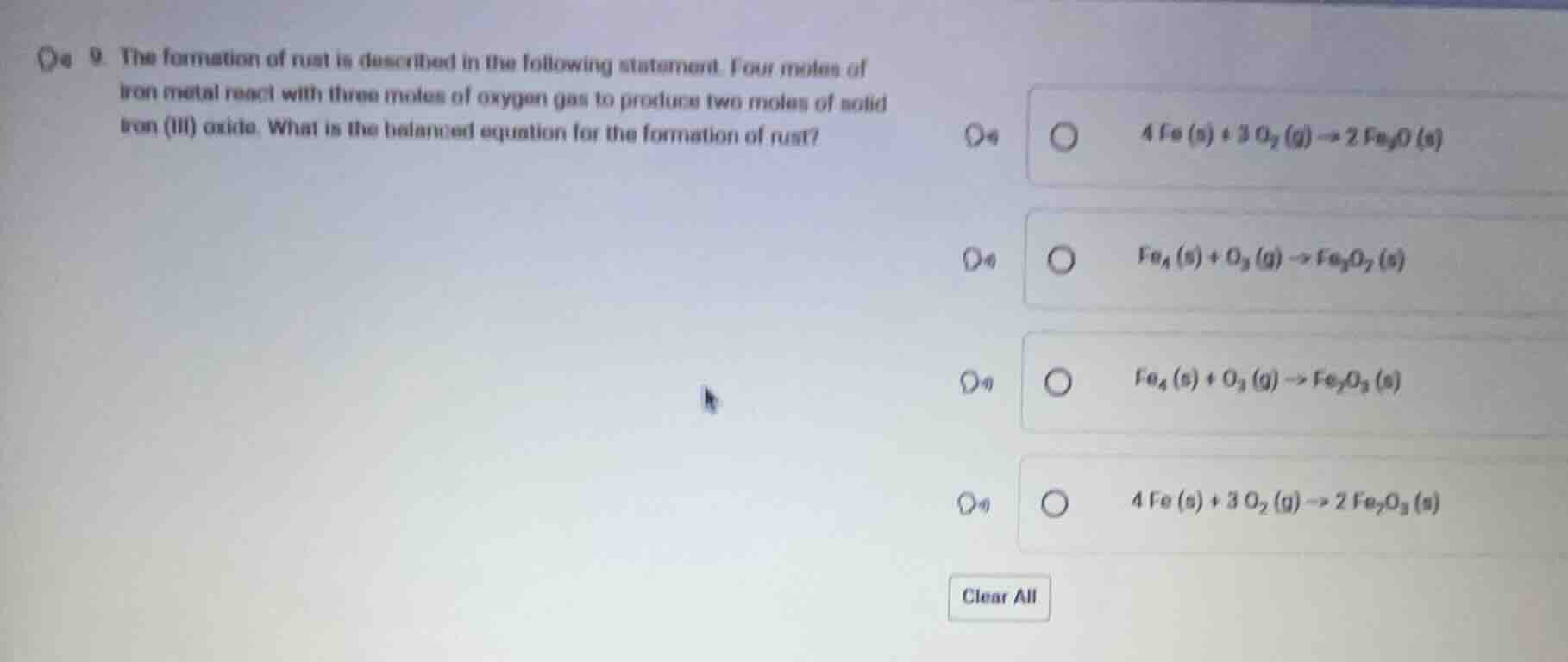
- the formation of rust is described in the following statement. four moles of iron metal react with three moles of oxygen gas to produce two moles of solid iron (iii) oxide. what is the balanced equation for the formation of rust?
4 fe (s) + 3 o₂ (g) → 2 fe₃o (s)
fe₄ (s) + o₃ (g) → fe₃o₂ (s)
fe₄ (s) + o₃ (g) → fe₂o₃ (s)
4 fe (s) + 3 o₂ (g) → 2 fe₂o₃ (s)
clear all
Step1: Identify reactants/products
Reactants: Fe (iron), $\text{O}_2$ (oxygen gas). Product: Iron(III) oxide, formula $\text{Fe}_2\text{O}_3$ (since Fe³⁺ and O²⁻ form neutral compound).
Step2: Match mole ratios
Problem states 4 mol Fe, 3 mol $\text{O}_2$ produce 2 mol product. Check atom balance:
- Fe: Left $4$, Right $2\times2=4$
- O: Left $3\times2=6$, Right $2\times3=6$
Step3: Verify correct option
Only the last option uses correct formula for iron(III) oxide and matches the given mole ratios with balanced atoms.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. $4 \text{ Fe (s)} + 3 \text{ O}_2 \text{ (g)}
ightarrow 2 \text{ Fe}_2\text{O}_3 \text{ (s)}$