QUESTION IMAGE
Question
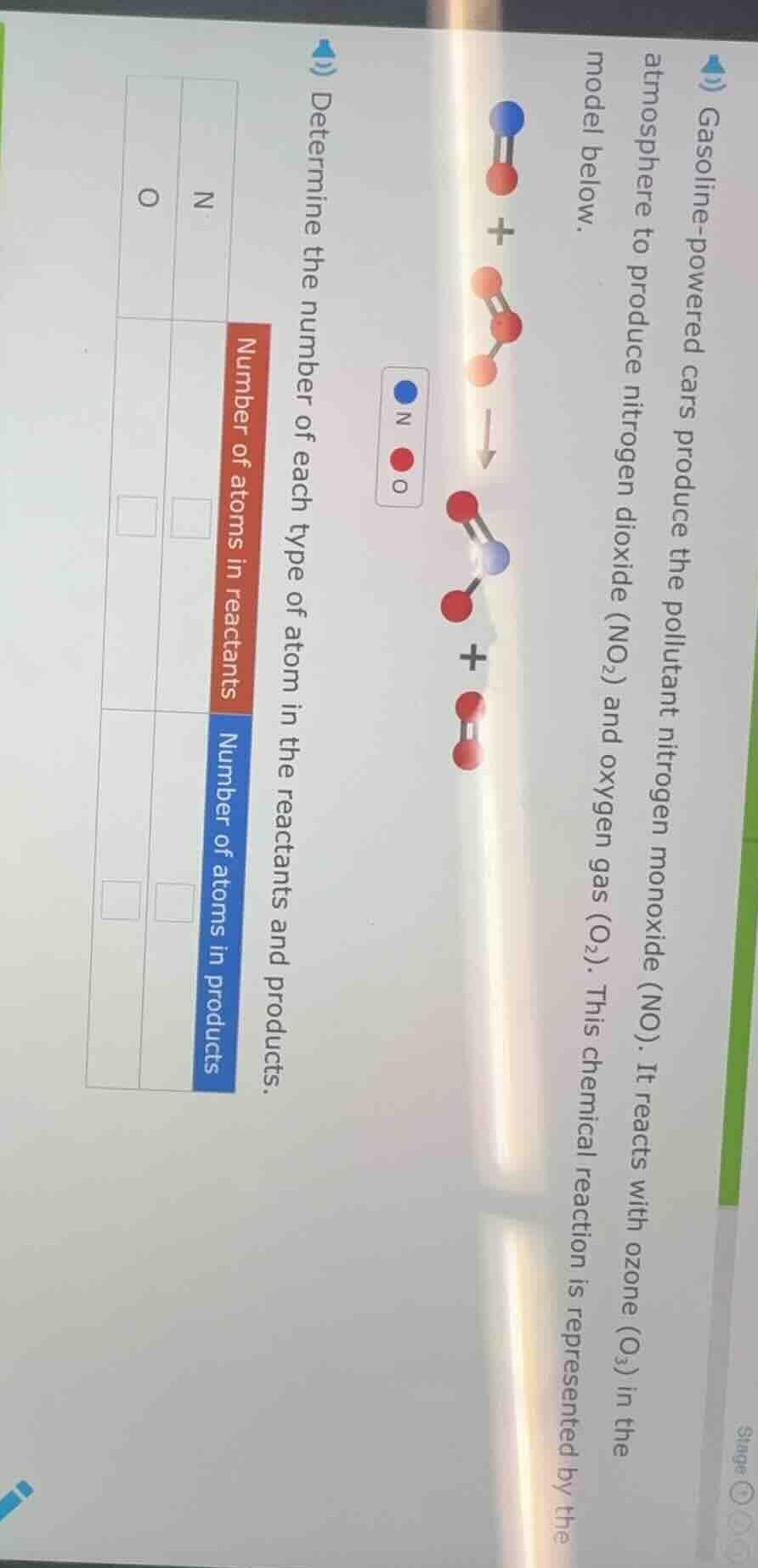
- gasoline-powered cars produce the pollutant nitrogen monoxide (no). it reacts with ozone (o₃) in the atmosphere to produce nitrogen dioxide (no₂) and oxygen gas (o₂). this chemical reaction is represented by the model below.determine the number of each type of atom in the reactants and products.number of atoms in reactantsn □o □number of atoms in productsn □o □(legend: blue = n, red = o)
Step1: Count reactant N atoms
Reactants: 1 NO molecule. Number of N atoms = 1
Step2: Count reactant O atoms
Reactants: 1 NO (1 O) + 1 O₃ (3 O). Total O atoms = $1 + 3 = 4$
Step3: Count product N atoms
Products: 1 NO₂ molecule. Number of N atoms = 1
Step4: Count product O atoms
Products: 1 NO₂ (2 O) + 1 O₂ (2 O). Total O atoms = $2 + 2 = 4$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Number of atoms in reactants | Number of atoms in products | |
|---|---|---|
| O | 4 | 4 |