QUESTION IMAGE
Question
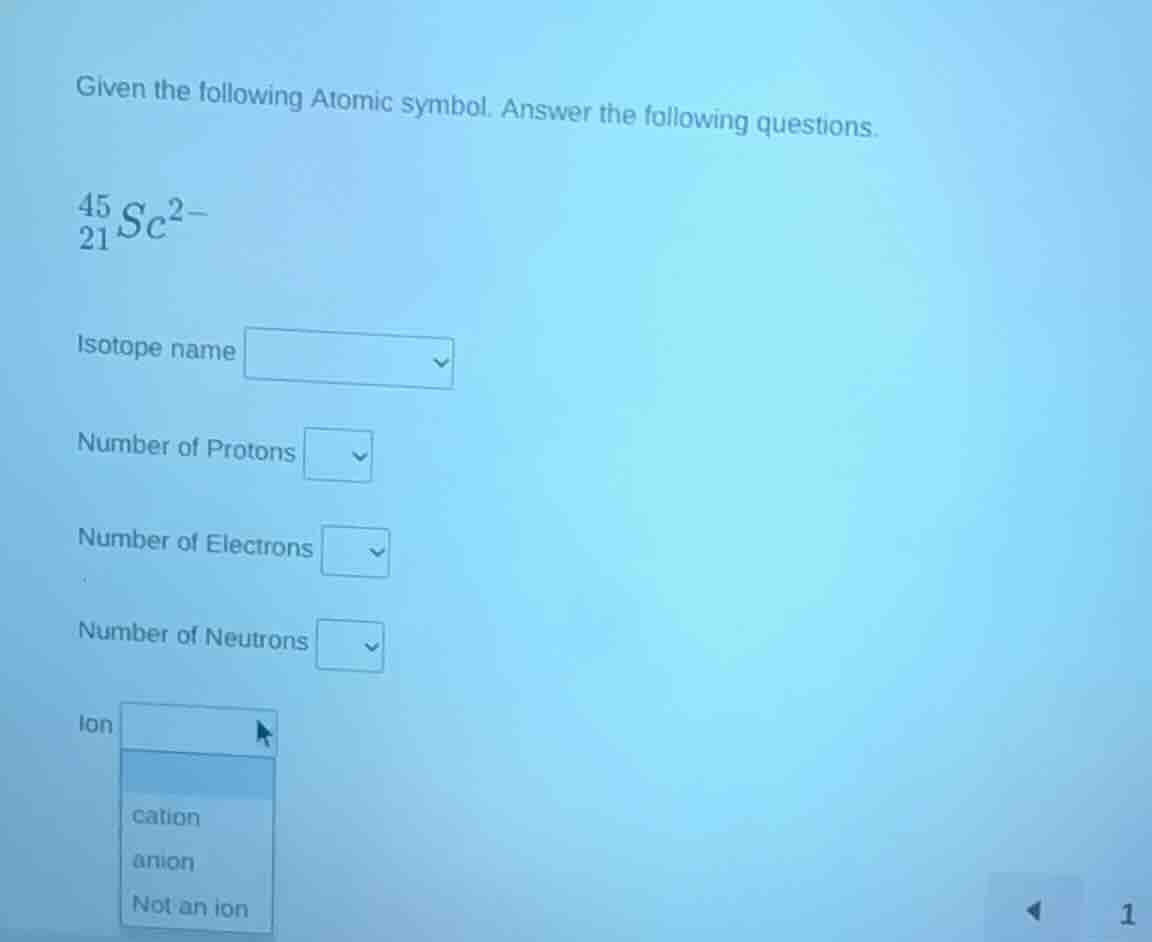
given the following atomic symbol. answer the following questions.
\\(_{21}^{45}sc^{2-}\\)
isotope name
number of protons
number of electrons
number of neutrons
ion
cation
anion
not an ion
Isotope Name
The atomic symbol is \(\ce{_{21}^{45}Sc^{2-}}\). The isotope name is formed by the element name (Scandium) followed by the mass number. So it's Scandium - 45.
The atomic number (the bottom number in the atomic symbol) is equal to the number of protons. Here, the atomic number is 21, so the number of protons is 21.
For a neutral atom, the number of electrons equals the number of protons. But this is an ion with a \(2^-\) charge, meaning it has gained 2 electrons. So number of electrons = number of protons + 2 = \(21 + 2=23\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Scandium - 45