QUESTION IMAGE
Question
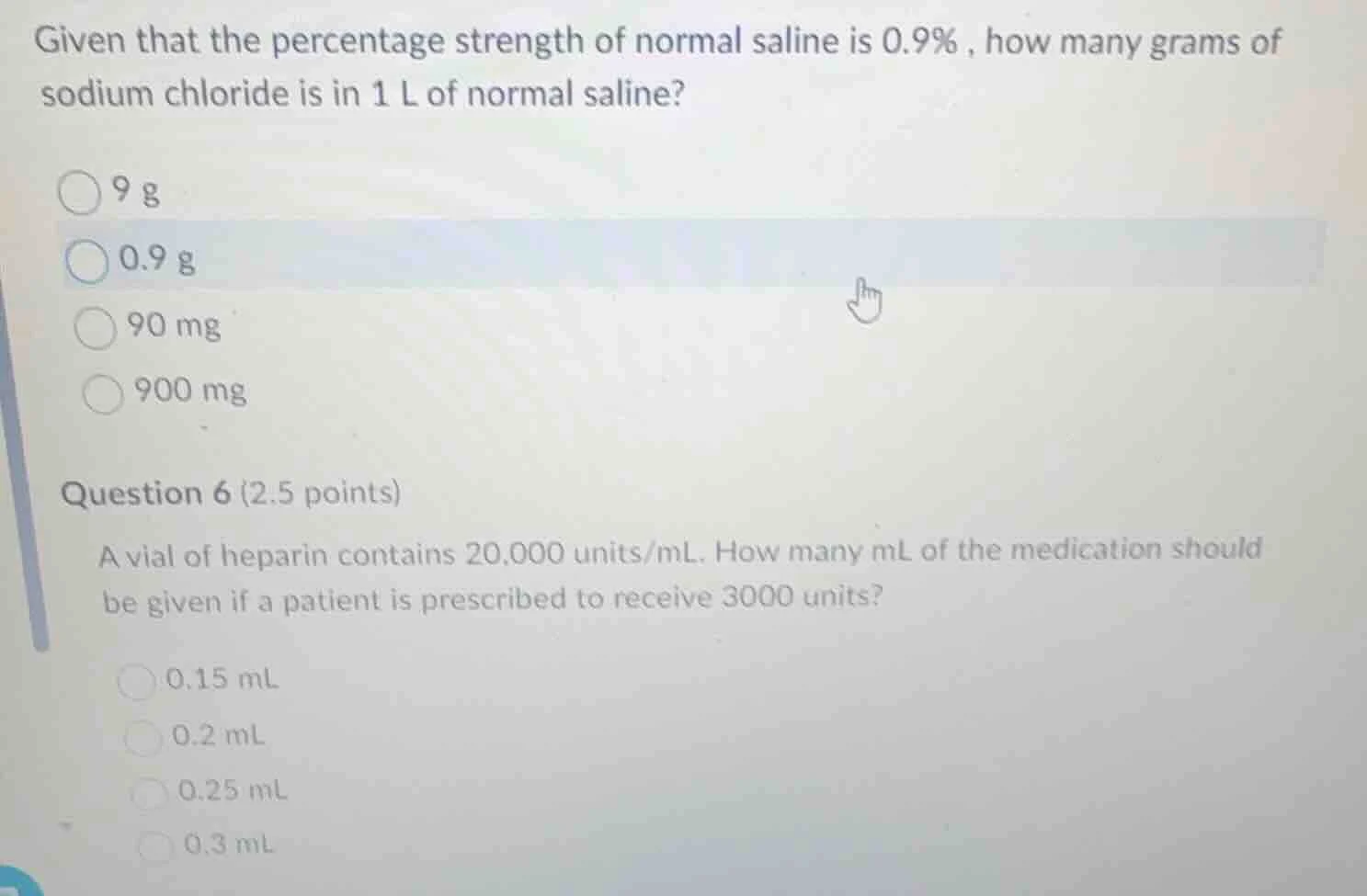
given that the percentage strength of normal saline is 0.9%, how many grams of sodium chloride is in 1 l of normal saline?
9 g
0.9 g
90 mg
900 mg
question 6 (2.5 points)
a vial of heparin contains 20,000 units/ml. how many ml of the medication should be given if a patient is prescribed to receive 3000 units?
0.15 ml
0.2 ml
0.25 ml
0.3 ml
First Question (Sodium Chloride in Normal Saline)
Step1: Understand the percentage strength
The percentage strength of 0.9% means 0.9 grams of sodium chloride per 100 mL of solution (since percentage strength is mass/volume in grams per 100 mL for solutions like saline).
Step2: Convert 1 L to mL
We know that 1 L = 1000 mL.
Step3: Calculate the amount of sodium chloride
If 100 mL has 0.9 g, then 1 mL has $\frac{0.9}{100}$ g. For 1000 mL (1 L), the amount is $\frac{0.9}{100} \times 1000 = 9$ g.
Step1: Recall the formula for dosage calculation
The formula is Volume (mL) = $\frac{\text{Desired Dose (units)}}{\text{Dose on Hand (units/mL)}}$.
Step2: Plug in the values
Desired Dose = 3000 units, Dose on Hand = 20,000 units/mL. So Volume = $\frac{3000}{20000} = 0.15$ mL.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
9 g (the first option: 9 g)