QUESTION IMAGE
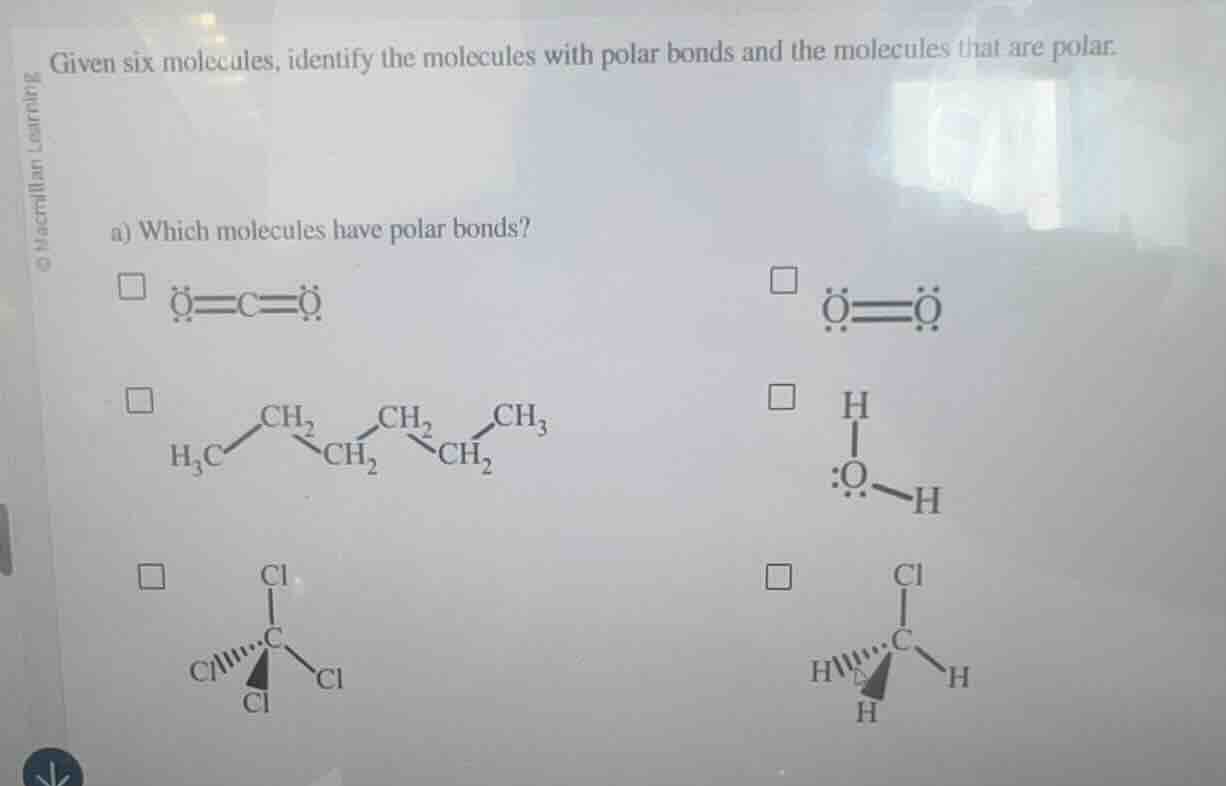
Question
given six molecules, identify the molecules with polar bonds and the molecules that are polar.
a) which molecules have polar bonds?
□ (ddot{o}=c=ddot{o}) □ (ddot{o}=ddot{o})
□ (ce{h3c-ch2-ch2-ch2-ch3}) □ (ce{h-o-h}) (with lone pairs on o)
□ (ce{ccl4}) (tetrahedral structure) □ (ce{ch3cl}) (tetrahedral structure with h and cl attached to c)
Brief Explanations
Polar bonds form when two bonded atoms have different electronegativities, creating a dipole.
- $\ce{O=C=O}$: C and O have different electronegativities, so C=O bonds are polar.
- $\ce{O=O}$: Bond between identical O atoms, no electronegativity difference, nonpolar bonds.
- $\ce{H3C(CH2)4CH3}$ (hexane): Only C-C and C-H bonds; electronegativity difference between C and H is negligible, nonpolar bonds.
- $\ce{H2O}$: O and H have a large electronegativity difference, so O-H bonds are polar.
- $\ce{CCl4}$: C and Cl have different electronegativities, so C-Cl bonds are polar.
- $\ce{CH3Cl}$: C and Cl, and C and H (small difference) have electronegativity differences, so C-Cl and C-H bonds are polar (C-Cl is strongly polar).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The molecules with polar bonds are:
- $\ce{O=C=O}$
- $\ce{H2O}$
- $\ce{CCl4}$
- $\ce{CH3Cl}$