QUESTION IMAGE
Question
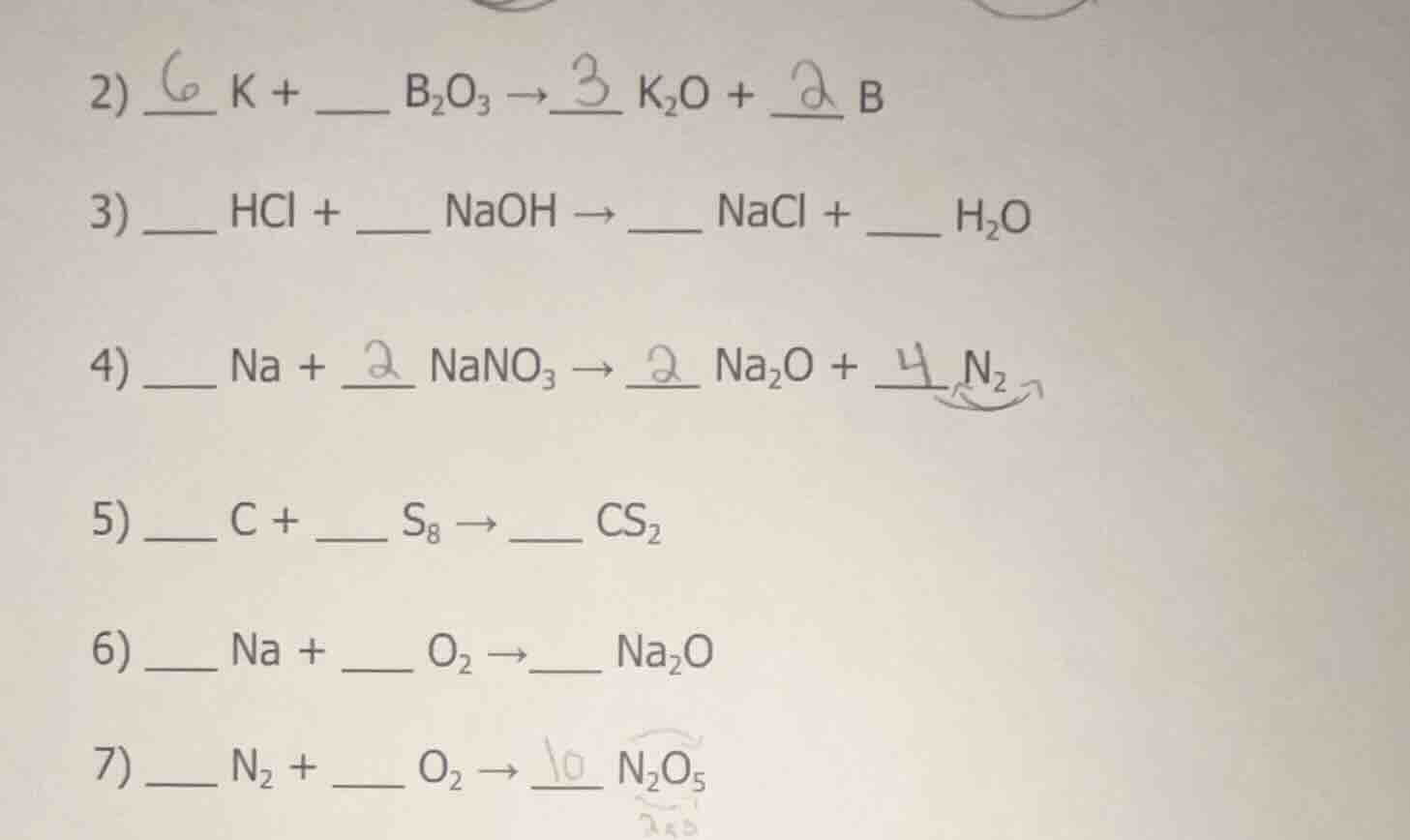
- \underline{6} k + \underline{\quad} b₂o₃ → \underline{3} k₂o + \underline{2} b\
- \underline{\quad} hcl + \underline{\quad} naoh → \underline{\quad} nacl + \underline{\quad} h₂o\
- \underline{\quad} na + \underline{2} nano₃ → \underline{2} na₂o + \underline{4} n₂\
- \underline{\quad} c + \underline{\quad} s₈ → \underline{\quad} cs₂\
- \underline{\quad} na + \underline{\quad} o₂ → \underline{\quad} na₂o\
- \underline{\quad} n₂ + \underline{\quad} o₂ → \underline{10} n₂o₅
Problem 2:
Step1: Balance O atoms.
On the left, \( B_2O_3 \) has 3 O. On the right, \( K_2O \) has 1 O. So put 3 in front of \( K_2O \) to make O: \( 3 \times 1 = 3 \). Now equation: \( \underline{\quad} K + \underline{\quad} B_2O_3
ightarrow \underline{3} K_2O + \underline{\quad} B \)
Step2: Balance K atoms.
\( 3 K_2O \) has \( 3 \times 2 = 6 \) K. So put 6 in front of K. Equation: \( \underline{6} K + \underline{\quad} B_2O_3
ightarrow \underline{3} K_2O + \underline{\quad} B \)
Step3: Balance B atoms.
\( B_2O_3 \) has 2 B. So put 2 in front of B. And check \( B_2O_3 \): only 1 needed. Final equation: \( \underline{6} K + \underline{1} B_2O_3
ightarrow \underline{3} K_2O + \underline{2} B \)
Step1: This is a neutralization reaction. HCl + NaOH → NaCl + H₂O. Check atoms:
H: 1 (HCl) + 1 (NaOH) = 2 (H₂O: 2 H). Cl: 1 (HCl) = 1 (NaCl). Na: 1 (NaOH) = 1 (NaCl). O: 1 (NaOH) = 1 (H₂O). So all atoms are balanced with 1 in each blank.
Step1: Balance N atoms.
Left: \( NaNO_3 \) has 1 N. Right: \( N_2 \) has 2 N. So put 2 in front of \( NaNO_3 \) to make N: \( 2 \times 1 = 2 \), matching \( N_2 \). Now equation: \( \underline{\quad} Na + \underline{2} NaNO_3
ightarrow \underline{\quad} Na_2O + \underline{\quad} N_2 \)
Step2: Balance O atoms.
\( 2 NaNO_3 \) has \( 2 \times 3 = 6 \) O. Right: \( Na_2O \) has 1 O. So put 6 in front of \( Na_2O \) (since \( 6 \times 1 = 6 \) O). Equation: \( \underline{\quad} Na + \underline{2} NaNO_3
ightarrow \underline{6} Na_2O + \underline{\quad} N_2 \)
Step3: Balance Na atoms.
Left: Na (let's say x) + \( 2 NaNO_3 \) (2 Na) = x + 2. Right: \( 6 Na_2O \) (12 Na). So x + 2 = 12 → x = 10. Put 10 in front of Na. Equation: \( \underline{10} Na + \underline{2} NaNO_3
ightarrow \underline{6} Na_2O + \underline{1} N_2 \) (Check N: 2, O: 6, Na: 10 + 2 = 12, and \( 6 Na_2O \) has 12 Na. Balanced.)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
6, 1, 3, 2