QUESTION IMAGE
Question
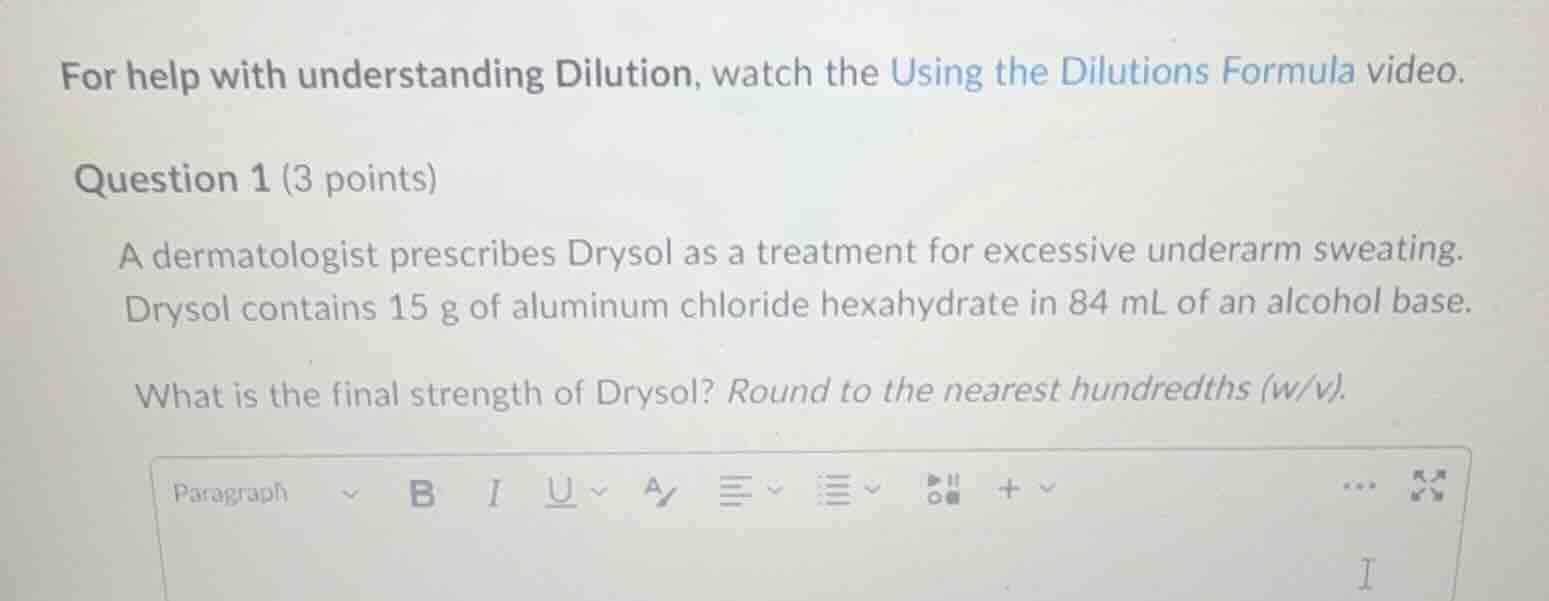
for help with understanding dilution, watch the using the dilutions formula video. question 1 (3 points) a dermatologist prescribes drysol as a treatment for excessive underarm sweating. drysol contains 15 g of aluminum chloride hexahydrate in 84 ml of an alcohol base. what is the final strength of drysol? round to the nearest hundredths (w/v).
Step1: Recall w/v formula
The weight/volume (w/v) percentage is calculated as $\frac{\text{mass of solute (g)}}{\text{volume of solution (mL)}} \times 100\%$.
Step2: Plug in values
Mass of solute (aluminum chloride hexahydrate) is 15 g, volume of solution is 84 mL. So we calculate $\frac{15}{84} \times 100\%$.
Step3: Perform calculation
$\frac{15}{84} \approx 0.17857$, then multiply by 100: $0.17857\times100 = 17.857\%$. Round to nearest hundredths: 17.86%.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
17.86%