QUESTION IMAGE
Question
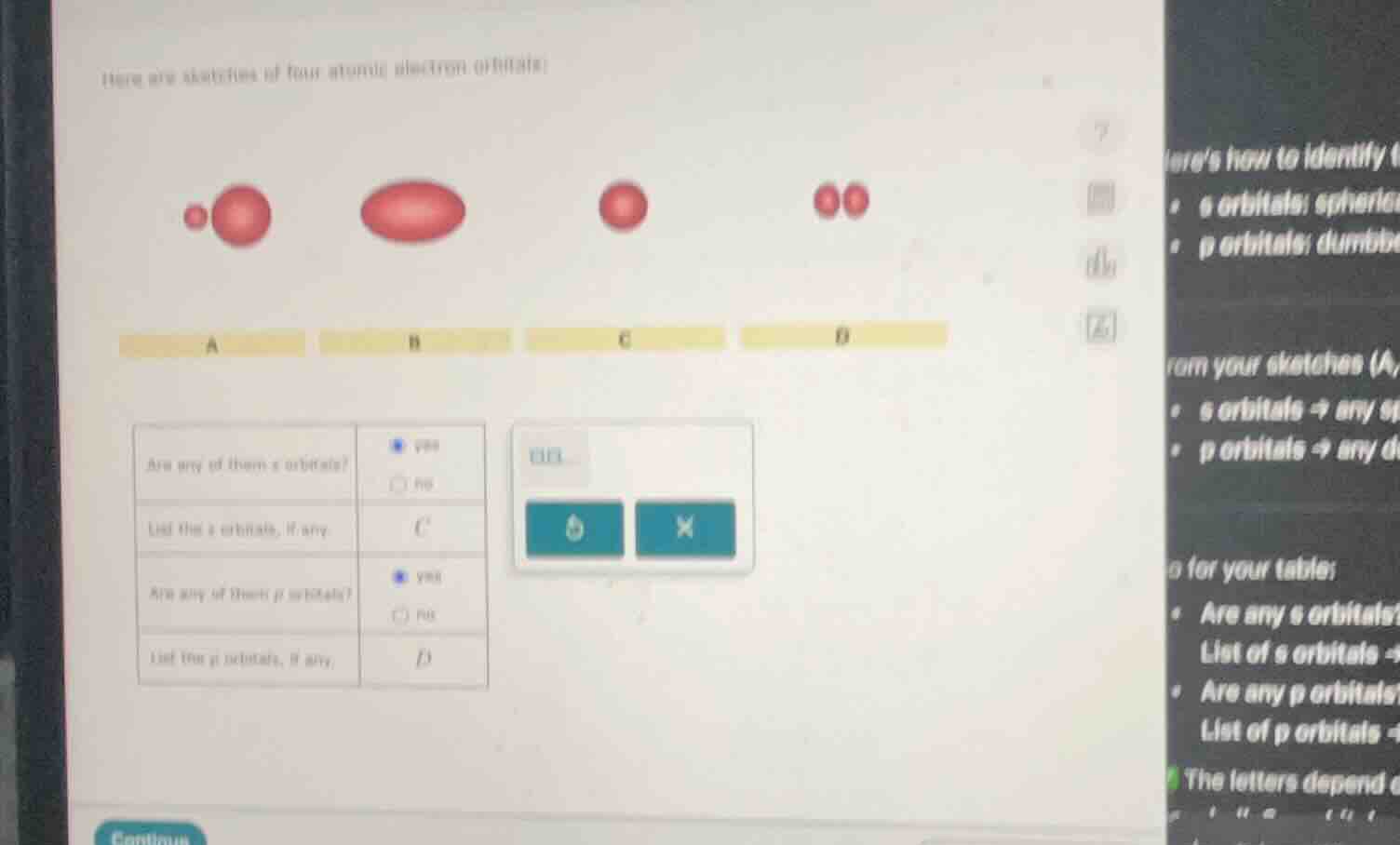
here are sketches of four atomic electron orbitals:
a
b
c
d
are any of them s orbitals?
yes
no
list the s orbitals, if any.
c
are any of them p orbitals?
yes
no
list the p orbitals, if any.
d
Step1: Recall s and p orbital shapes
s orbitals are spherical (symmetric, single sphere), p orbitals are dumbbell - shaped (or have two lobes, can be in different orientations like along axes, or have two distinct regions).
Step2: Analyze each orbital (A, B, C, D)
- Orbital C: It is a single sphere, so it matches the shape of an s orbital.
- Orbital D: It has two distinct lobes (the two red spheres close together), which matches the shape of a p orbital. Orbitals A and B: A has two spheres of different sizes (not s or typical p), B is an oval - like shape (not s or typical p).
Step3: Answer the questions
- "Are any of them s orbitals?": Yes, because C is an s orbital.
- "List the s orbitals, if any": C (since C is spherical, s - orbital shape).
- "Are any of them p orbitals?": Yes, because D has two lobes (p - orbital shape).
- "List the p orbitals, if any": D (since D has two lobes, characteristic of p orbitals).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Are any of them s orbitals? $\boldsymbol{Yes}$
- List the s orbitals, if any: $\boldsymbol{C}$
- Are any of them p orbitals? $\boldsymbol{Yes}$
- List the p orbitals, if any: $\boldsymbol{D}$