QUESTION IMAGE
Question
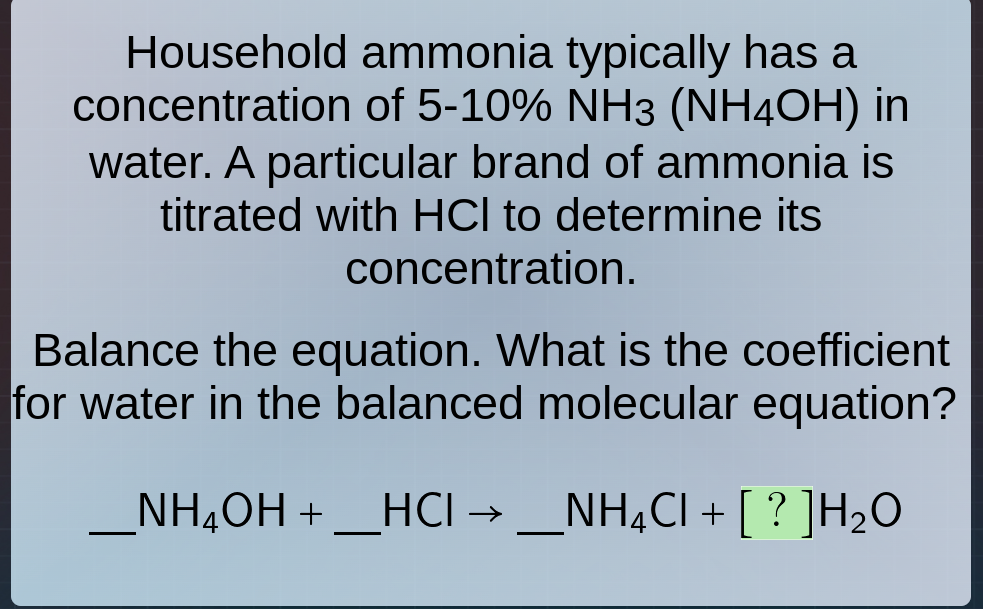
household ammonia typically has a concentration of 5-10% nh₃ (nh₄oh) in water. a particular brand of ammonia is titrated with hcl to determine its concentration. balance the equation. what is the coefficient for water in the balanced molecular equation? nh₄oh + hcl → __nh₄cl + ?h₂o
Step1: Analyze reactants and products
Reactants: $\ce{NH4OH}$ (ammonium hydroxide) and $\ce{HCl}$ (hydrochloric acid). Products: $\ce{NH4Cl}$ (ammonium chloride) and $\ce{H2O}$ (water).
Step2: Balance atoms
- For $\ce{N}$: 1 in $\ce{NH4OH}$, 1 in $\ce{NH4Cl}$ → balanced.
- For $\ce{Cl}$: 1 in $\ce{HCl}$, 1 in $\ce{NH4Cl}$ → balanced.
- For $\ce{H}$: In $\ce{NH4OH}$: 4 (from $\ce{NH4^+}$) + 1 (from $\ce{OH^-}$) = 5. In $\ce{HCl}$: 1. Total H on left: 5 + 1 = 6. On right: In $\ce{NH4Cl}$: 4, in $\ce{H2O}$: let coefficient be $x$, so 4 + 2x. For $\ce{O}$: 1 in $\ce{NH4OH}$, $x$ in $\ce{H2O}$. Wait, simpler: this is an acid-base reaction (neutralization). $\ce{OH^- + H^+ -> H2O}$. So $\ce{NH4OH + HCl -> NH4Cl + H2O}$. Let's check:
- $\ce{NH4OH}$: $\ce{NH4^+ + OH^-}$
- $\ce{HCl}$: $\ce{H^+ + Cl^-}$
- Reaction: $\ce{OH^- + H^+ -> H2O}$, and $\ce{NH4^+ + Cl^- -> NH4Cl}$. So combining: $\ce{NH4OH + HCl -> NH4Cl + H2O}$. Now check each atom:
- $\ce{N}$: 1 on left, 1 on right.
- $\ce{Cl}$: 1 on left, 1 on right.
- $\ce{O}$: 1 on left (in $\ce{OH^-}$), 1 on right (in $\ce{H2O}$).
- $\ce{H}$: In $\ce{NH4OH}$: 4 (from $\ce{NH4^+}$) + 1 (from $\ce{OH^-}$) = 5; in $\ce{HCl}$: 1. Total H left: 6. On right: in $\ce{NH4Cl}$: 4, in $\ce{H2O}$: 2 (since coefficient 1, 2*1=2). 4 + 2 = 6. Balanced. So coefficients: 1 for $\ce{NH4OH}$, 1 for $\ce{HCl}$, 1 for $\ce{NH4Cl}$, 1 for $\ce{H2O}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1