QUESTION IMAGE
Question
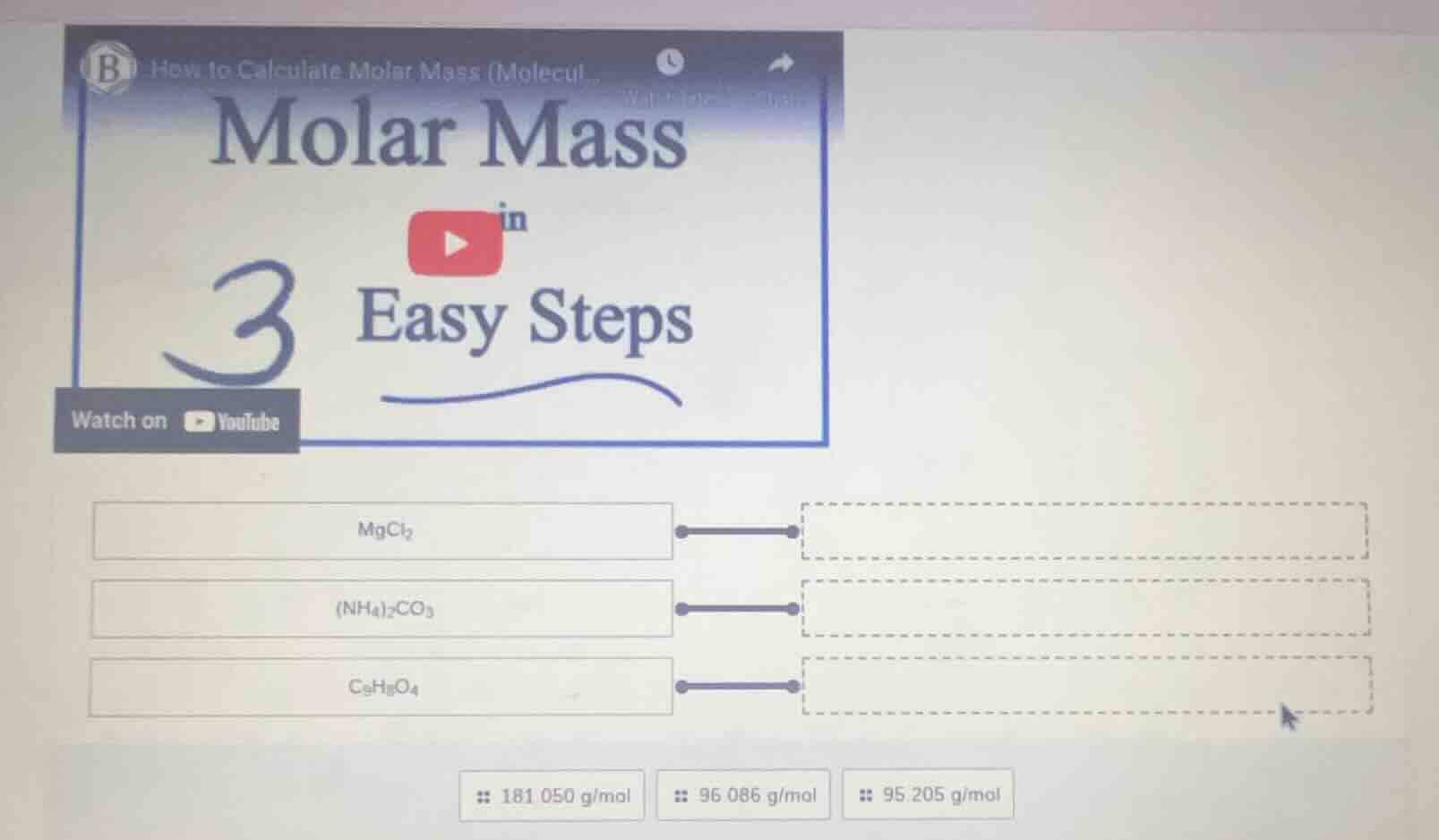
how to calculate molar mass (molecul... molar mass in 3 easy steps watch on youtube mgcl₂ (nh₄)₂co₃ c₆h₅o₄ 181.050 g/mol 96.086 g/mol 95.205 g/mol
For \( \boldsymbol{\ce{MgCl2}} \):
Step1: Find atomic masses
Atomic mass of \( \ce{Mg} = 24.305 \, \text{g/mol} \), atomic mass of \( \ce{Cl} = 35.453 \, \text{g/mol} \).
Step2: Calculate molar mass
\( \ce{MgCl2} \) has 1 \( \ce{Mg} \) and 2 \( \ce{Cl} \).
Molar mass \( = 24.305 + 2\times35.453 = 24.305 + 70.906 = 95.211 \, \text{g/mol} \) (close to \( 95.205 \, \text{g/mol} \)).
For \( \boldsymbol{\ce{(NH4)2CO3}} \):
Step1: Find atomic masses
Atomic mass of \( \ce{N} = 14.007 \, \text{g/mol} \), \( \ce{H} = 1.008 \, \text{g/mol} \), \( \ce{C} = 12.011 \, \text{g/mol} \), \( \ce{O} = 16.00 \, \text{g/mol} \).
Step2: Calculate molar mass
\( \ce{(NH4)2CO3} \) has 2 \( \ce{N} \), 8 \( \ce{H} \), 1 \( \ce{C} \), 3 \( \ce{O} \).
Molar mass \( = 2\times(14.007 + 4\times1.008) + 12.011 + 3\times16.00 \)
\( = 2\times(14.007 + 4.032) + 12.011 + 48.00 \)
\( = 2\times18.039 + 12.011 + 48.00 \)
\( = 36.078 + 12.011 + 48.00 = 96.089 \, \text{g/mol} \) (close to \( 96.086 \, \text{g/mol} \)).
For \( \boldsymbol{\ce{C9H9O4}} \):
Step1: Find atomic masses
Atomic mass of \( \ce{C} = 12.011 \, \text{g/mol} \), \( \ce{H} = 1.008 \, \text{g/mol} \), \( \ce{O} = 16.00 \, \text{g/mol} \).
Step2: Calculate molar mass
\( \ce{C9H9O4} \) has 9 \( \ce{C} \), 9 \( \ce{H} \), 4 \( \ce{O} \).
Molar mass \( = 9\times12.011 + 9\times1.008 + 4\times16.00 \)
\( = 108.099 + 9.072 + 64.00 = 181.171 \, \text{g/mol} \) (close to \( 181.050 \, \text{g/mol} \)).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- \( \ce{MgCl2} \) matches \( 95.205 \, \text{g/mol} \)
- \( \ce{(NH4)2CO3} \) matches \( 96.086 \, \text{g/mol} \)
- \( \ce{C9H9O4} \) matches \( 181.050 \, \text{g/mol} \)