QUESTION IMAGE
Question
how fda regulates artificial intelligence in medical products
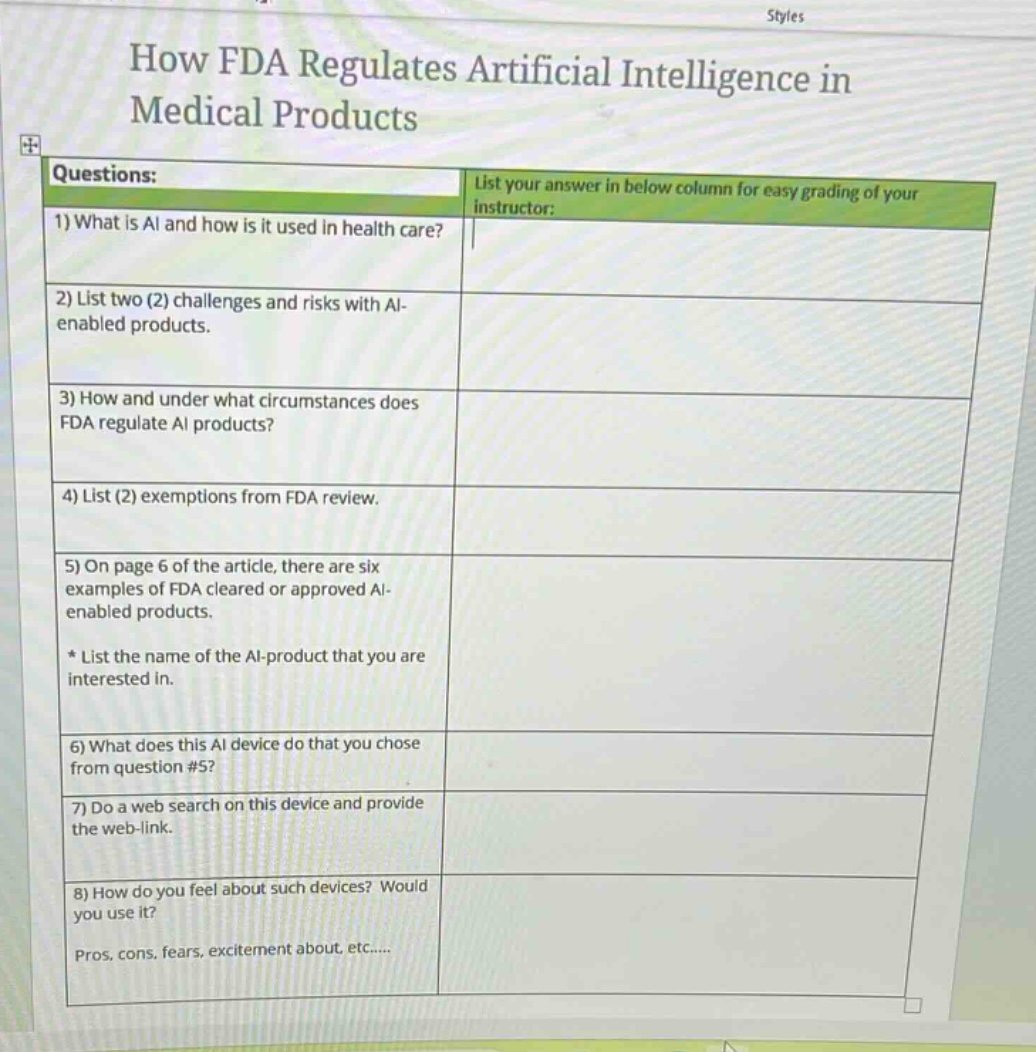
questions:
- what is ai and how is it used in health care?
- list two (2) challenges and risks with ai - enabled products.
- how and under what circumstances does fda regulate ai products?
- list (2) exemptions from fda review.
- on page 6 of the article, there are six examples of fda cleared or approved ai - enabled products.
- list the name of the ai - product that you are interested in.
- what does this ai device do that you chose from question #5?
- do a web search on this device and provide the web - link.
- how do you feel about such devices? would you use it?
pros, cons, fears, excitement about, etc......
list your answer in below column for easy grading of your instructor:
To answer these questions, we'll address each one based on general knowledge about AI in healthcare and FDA regulations (and you may need to refer to the specific article for questions 4 - 8 details):
1) What is AI and how is it used in health care?
AI (Artificial Intelligence) refers to computer systems that mimic human intelligence (e.g., learning, problem - solving). In healthcare, it’s used for:
- Diagnosis: Analyzing medical images (like X - rays, MRIs) to detect diseases (e.g., cancer, fractures).
- Treatment Planning: Personalizing treatment plans (e.g., cancer therapies) based on patient data.
- Drug Discovery: Identifying new drug candidates and predicting drug - disease interactions.
- Administrative Tasks: Streamlining patient records, scheduling, and billing.
- Bias & Fairness: AI models trained on non - representative data may produce biased results (e.g., misdiagnosing patients from underrepresented ethnic groups).
- Lack of Transparency (Black Box): Some AI algorithms (e.g., deep learning) are hard to interpret, making it difficult to trust or audit decisions (e.g., why a diagnosis was made).
The FDA regulates AI - enabled medical products (like software, devices) as medical devices (Class I, II, or III, based on risk). Circumstances:
- If the AI is a medical device (e.g., used for diagnosis, treatment, or patient management), it must meet FDA’s premarket review (e.g., 510(k) for low - risk, PMA for high - risk) and postmarket surveillance requirements.
- For “software as a medical device” (SaMD), the FDA evaluates its intended use, clinical validity, and cybersecurity.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
AI is computer - based intelligence mimicking human thinking (learning, problem - solving). In healthcare, it’s used for medical imaging diagnosis (e.g., detecting tumors in X - rays), personalized treatment planning (e.g., cancer care), drug discovery, and administrative tasks (e.g., patient record management).