QUESTION IMAGE
Question
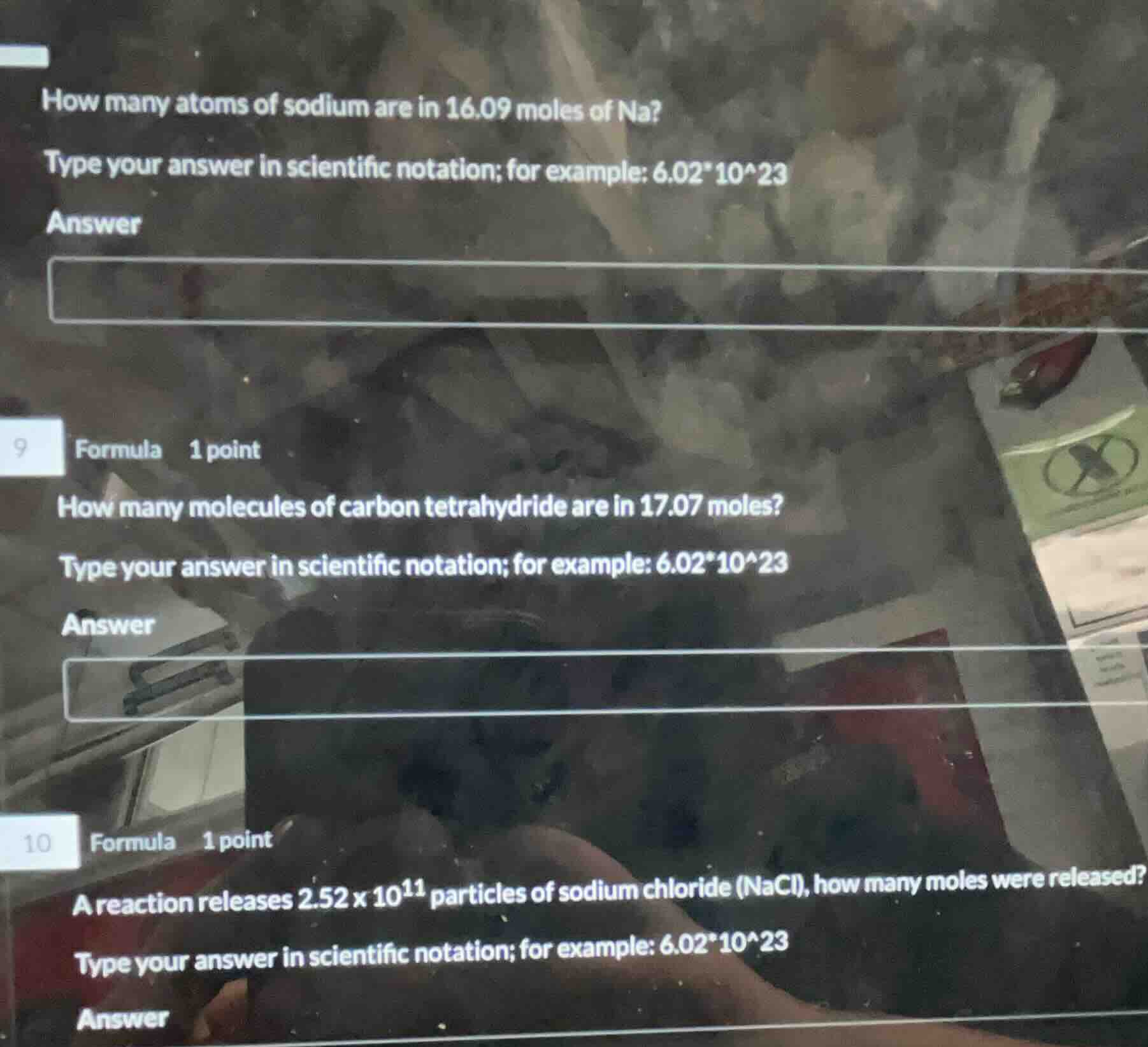
how many atoms of sodium are in 16.09 moles of na?
type your answer in scientific notation; for example: 6.02*10^23
answer
9 formula 1 point
how many molecules of carbon tetrahydride are in 17.07 moles?
type your answer in scientific notation; for example: 6.02*10^23
answer
10 formula 1 point
a reaction releases $2.52 \times 10^{11}$ particles of sodium chloride (nacl), how many moles were released?
type your answer in scientific notation; for example: 6.02*10^23
answer
Step1: Use Avogadro's number for Na atoms
Number of atoms = Moles × Avogadro's number
$\text{Atoms of Na} = 16.09 \times 6.02 \times 10^{23}$
$\text{Atoms of Na} = 9.68618 \times 10^{24} \approx 9.69 \times 10^{24}$
Step2: Use Avogadro's number for CH₄ molecules
Number of molecules = Moles × Avogadro's number
$\text{Molecules of } \text{CH}_4 = 17.07 \times 6.02 \times 10^{23}$
$\text{Molecules of } \text{CH}_4 = 1.027614 \times 10^{25} \approx 1.03 \times 10^{25}$
Step3: Calculate moles of NaCl from particles
Moles = $\frac{\text{Number of particles}}{\text{Avogadro's number}}$
$\text{Moles of NaCl} = \frac{2.52 \times 10^{11}}{6.02 \times 10^{23}}$
$\text{Moles of NaCl} \approx 4.186 \times 10^{-13} \approx 4.19 \times 10^{-13}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $9.69 \times 10^{24}$
- $1.03 \times 10^{25}$
- $4.19 \times 10^{-13}$