QUESTION IMAGE
Question
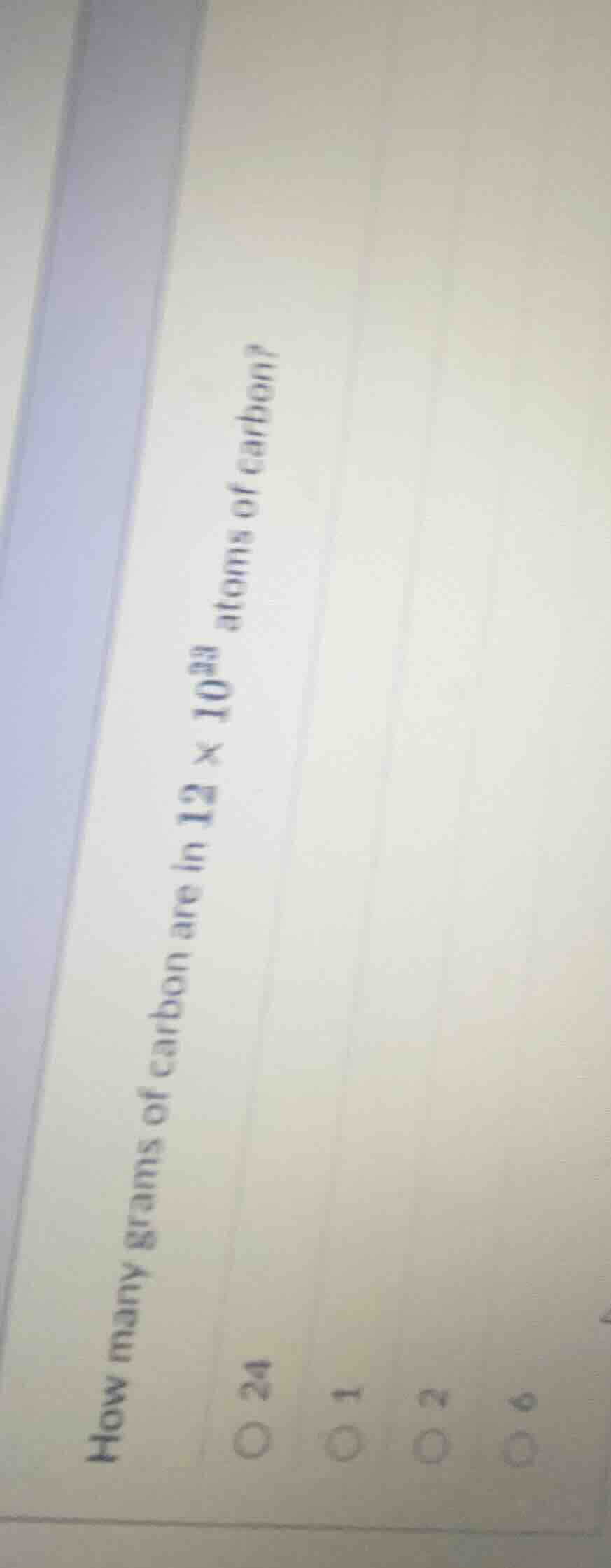
how many grams of carbon are in $12 \times 10^{24}$ atoms of carbon?
\bigcirc 24
\bigcirc 1
\bigcirc 2
\bigcirc 6
Step1: Recall Avogadro's number
Avogadro's number $N_A = 6.022 \times 10^{23} \text{ atoms/mol}$
Step2: Calculate moles of carbon
$\text{Moles} = \frac{\text{Number of atoms}}{N_A} = \frac{12 \times 10^{23}}{6.022 \times 10^{23}} \approx 1.993 \text{ mol}$
Step3: Use molar mass of C
Molar mass of C is $12 \text{ g/mol}$. $\text{Mass} = \text{Moles} \times \text{Molar Mass}$
$\text{Mass} = 1.993 \times 12 \approx 24 \text{ g}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
24