QUESTION IMAGE
Question
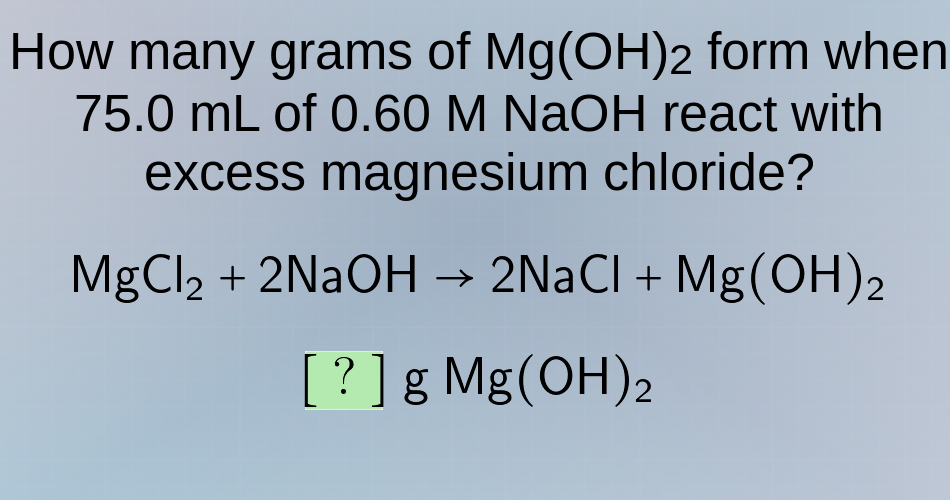
how many grams of mg(oh)₂ form when 75.0 ml of 0.60 m naoh react with excess magnesium chloride? mgcl₂ + 2naoh → 2nacl + mg(oh)₂ ? g mg(oh)₂
Step1: Find moles of NaOH
Molarity formula: $M = \frac{n}{V}$, so $n = M \times V$. Convert volume to liters: $75.0\ mL = 0.075\ L$.
$n_{NaOH} = 0.60\ M \times 0.075\ L = 0.045\ mol$.
Step2: Relate moles of NaOH to Mg(OH)₂
From reaction: $2\ mol\ NaOH$ produce $1\ mol\ Mg(OH)_2$.
Mole ratio: $\frac{1\ mol\ Mg(OH)_2}{2\ mol\ NaOH}$.
$n_{Mg(OH)_2} = 0.045\ mol\ NaOH \times \frac{1\ mol\ Mg(OH)_2}{2\ mol\ NaOH} = 0.0225\ mol$.
Step3: Calculate molar mass of Mg(OH)₂
Molar mass: $Mg (24.31) + 2\times O (16.00) + 2\times H (1.01) = 24.31 + 32.00 + 2.02 = 58.33\ g/mol$.
Step4: Find mass of Mg(OH)₂
Mass formula: $m = n \times M$.
$m_{Mg(OH)_2} = 0.0225\ mol \times 58.33\ g/mol \approx 1.31\ g$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1.31