QUESTION IMAGE
Question
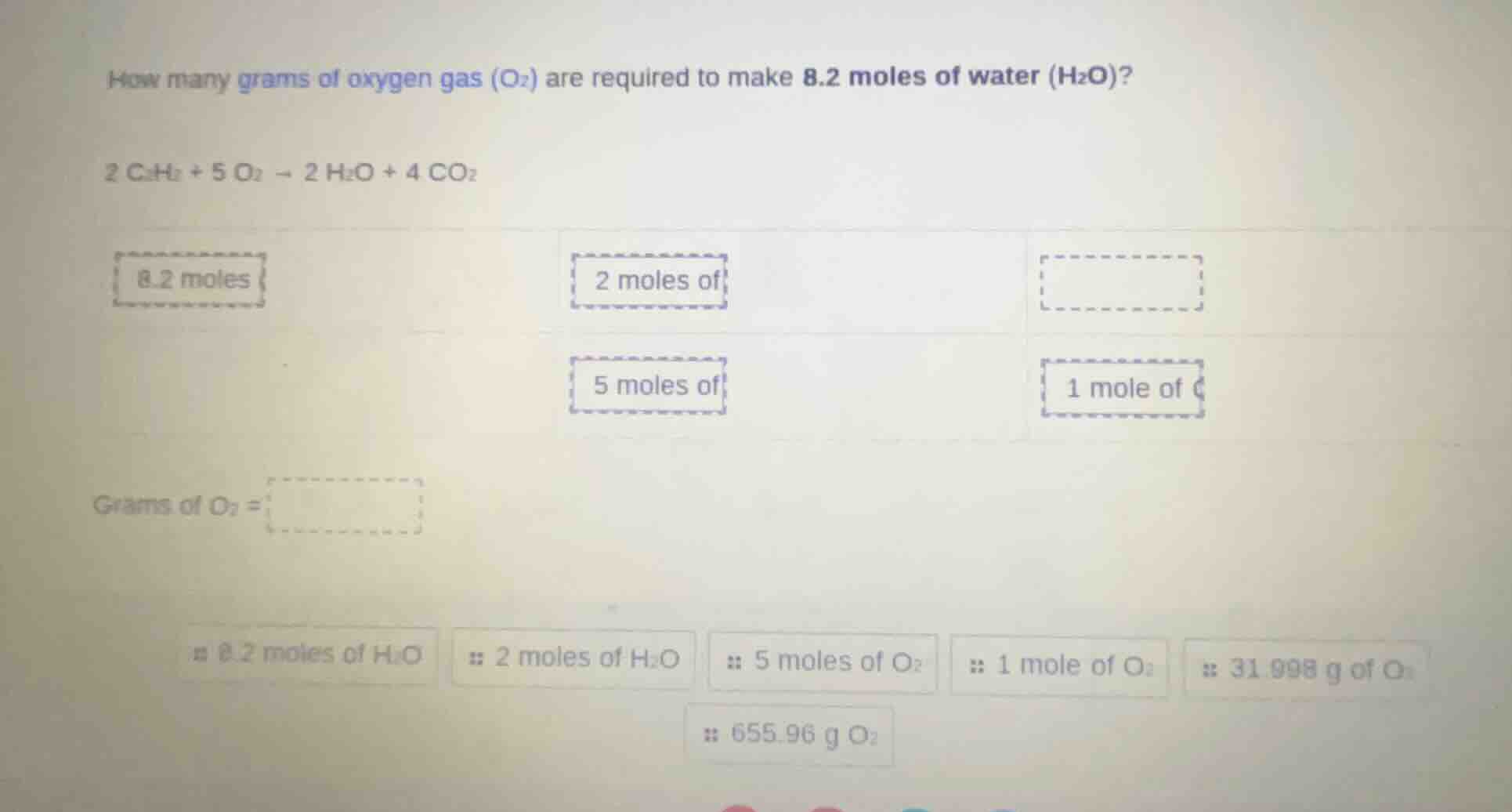
how many grams of oxygen gas (o₂) are required to make 8.2 moles of water (h₂o)?
2 c₂h₂ + 5 o₂ → 2 h₂o + 4 co₂
grams of o₂ =
Step1: Determine mole ratio of \(O_2\) to \(H_2O\)
From the reaction \(2 C_2H_2 + 5 O_2
ightarrow 2 H_2O + 4 CO_2\), the mole ratio of \(O_2\) to \(H_2O\) is \(\frac{5\space moles\space O_2}{2\space moles\space H_2O}\).
Step2: Calculate moles of \(O_2\)
Given moles of \(H_2O = 8.2\space moles\), moles of \(O_2 = 8.2\space moles\space H_2O\times\frac{5\space moles\space O_2}{2\space moles\space H_2O}\)
\[= \frac{8.2\times5}{2}\space moles\space O_2 = 20.5\space moles\space O_2\]
Step3: Convert moles of \(O_2\) to grams
Molar mass of \(O_2\) is \(31.998\space g/mol\). Mass of \(O_2 = 20.5\space moles\times31.998\space g/mol\)
\[= 20.5\times31.998 = 655.959\space g\approx656.0\space g\] (or as per the given \(31.998\) and calculation, it matches the provided \(655.96\space g\))
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The mass of oxygen gas required is \(\boldsymbol{655.96\space g}\) (or approximately \(656\space g\))