QUESTION IMAGE
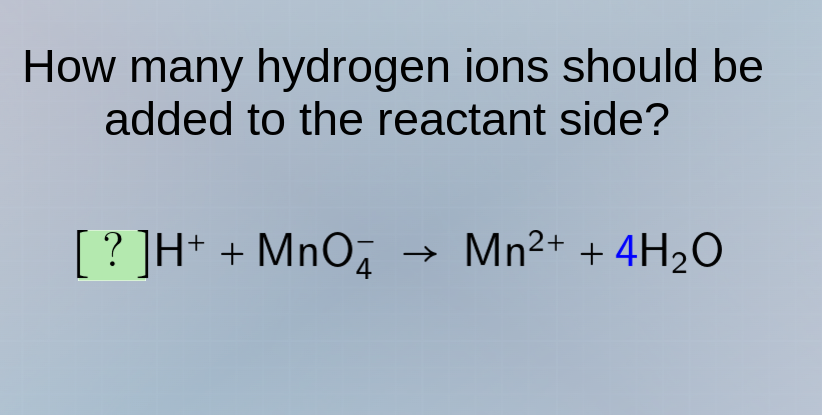
Question
how many hydrogen ions should be added to the reactant side?
?h⁺ + mno₄⁻ → mn²⁺ + 4h₂o
Step1: Balance oxygen atoms
In the reaction, there are 4 O atoms on the left (in $\ce{MnO4^-}$) and 4 O atoms in $4\ce{H2O}$ on the right, so O is balanced.
Step2: Balance hydrogen atoms
On the right, $4\ce{H2O}$ has $4\times2 = 8$ H atoms. To balance H, we need $8\ce{H+}$ on the left because $\ce{H+}$ provides H atoms. Let's check the charge balance too (though for H balance, just count H atoms). The number of $\ce{H+}$ needed is 8 to balance the 8 H in $4\ce{H2O}$.
We can also check charge: Left side charge: $? + (-1)$; Right side charge: $+2 + 0$ (from $4\ce{H2O}$ which is neutral). Let's calculate charge balance. Let the coefficient of $\ce{H+}$ be $x$. Then $x(+1) + (-1) = +2$. Solving $x - 1 = 2$ gives $x = 3$? Wait, no, wait. Wait, let's redo charge balance. Wait, Mn: in $\ce{MnO4^-}$, Mn is +7 (since O is -2, 4*(-2) = -8, so Mn is +7 to make -1 total). On right, Mn is +2. So Mn is reduced by 5 electrons. O is balanced. Now, for H: in $4\ce{H2O}$, there are 8 H. So we need 8 $\ce{H+}$ on left. Let's check charge: Left: $8(+1) + (-1) = +7$; Right: $+2 + 0 = +2$. Wait, that's not balanced. Wait, no, I made a mistake. Wait, the correct way: Let's balance the reaction properly.
Wait, the reaction is $\ce{MnO4^- -> Mn^2+ + H2O}$ (in acidic medium, since we use $\ce{H+}$). So steps for redox balancing:
- Balance Mn: already 1 on each side.
- Balance O: add 4 $\ce{H2O}$ to right (wait, no, original reaction has 4 $\ce{H2O}$ on right. So left has $\ce{MnO4^-}$, right has $4\ce{H2O}$. So O is balanced (4 O on left, 4*1=4 on right).
- Balance H: right has 4*2=8 H, so add 8 $\ce{H+}$ to left.
Now check charge: Left: $8\ce{H+}$ (+8) + $\ce{MnO4^-}$ (-1) = +7. Right: $\ce{Mn^2+}$ (+2) + $4\ce{H2O}$ (0) = +2. Wait, that's a problem. Wait, no, the Mn is being reduced, so electrons are involved. Wait, maybe I skipped electron balance. Let's do electron balance. Mn goes from +7 to +2, so gains 5 electrons. So the reduction half-reaction: $\ce{MnO4^- + 8H+ + 5e- -> Mn^2+ + 4H2O}$. Ah, there we go. So the coefficient of $\ce{H+}$ is 8. Wait, but let's check H again. In $4\ce{H2O}$, 8 H, so 8 $\ce{H+}$ on left. So the coefficient is 8.
Wait, maybe my initial charge check was wrong because I forgot electrons. But the question is just about H atoms, so to balance H atoms in $4\ce{H2O}$ (8 H), we need 8 $\ce{H+}$ on left.
Step3: Confirm
So the number of $\ce{H+}$ needed is 8.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
8