QUESTION IMAGE
Question
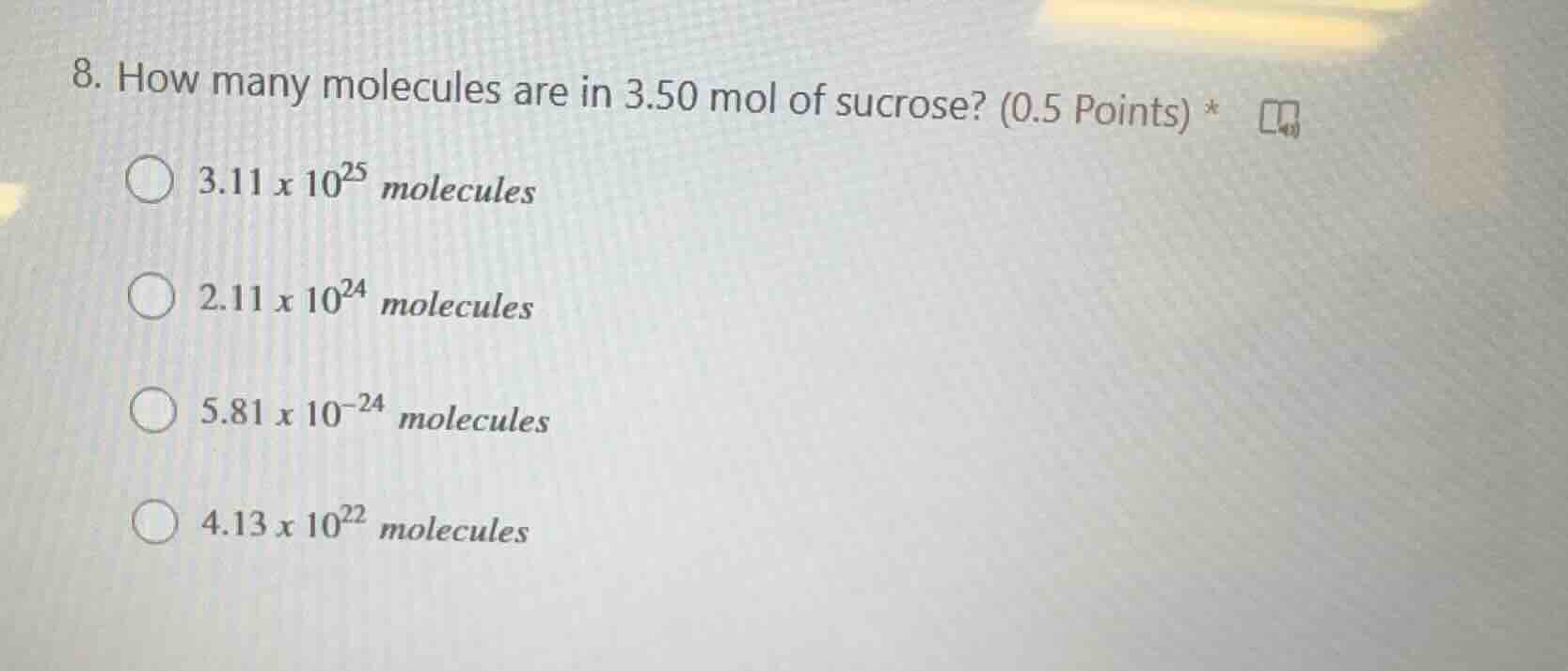
- how many molecules are in 3.50 mol of sucrose? (0.5 points) *
3.11 x 10²⁵ molecules
2.11 x 10²⁴ molecules
5.81 x 10⁻²⁴ molecules
4.13 x 10²² molecules
Step1: Recall Avogadro's number
Avogadro's number is \( 6.022\times 10^{23} \) molecules per mole. This means that 1 mole of any substance contains \( 6.022\times 10^{23} \) molecules.
Step2: Calculate the number of molecules
To find the number of molecules in 3.50 moles of sucrose, we multiply the number of moles by Avogadro's number. The formula is:
\[
\text{Number of molecules} = \text{Moles} \times \text{Avogadro's number}
\]
Substituting the values:
\[
\text{Number of molecules} = 3.50\ \text{mol} \times 6.022\times 10^{23}\ \frac{\text{molecules}}{\text{mol}}
\]
\[
\text{Number of molecules} = 3.50\times 6.022\times 10^{23}
\]
\[
3.50\times 6.022 = 21.077
\]
\[
\text{Number of molecules} = 21.077\times 10^{23} = 2.1077\times 10^{24}\approx 2.11\times 10^{24}\ \text{molecules}
\]
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2.11 x 10²⁴ molecules (the option with this value)