QUESTION IMAGE
Question
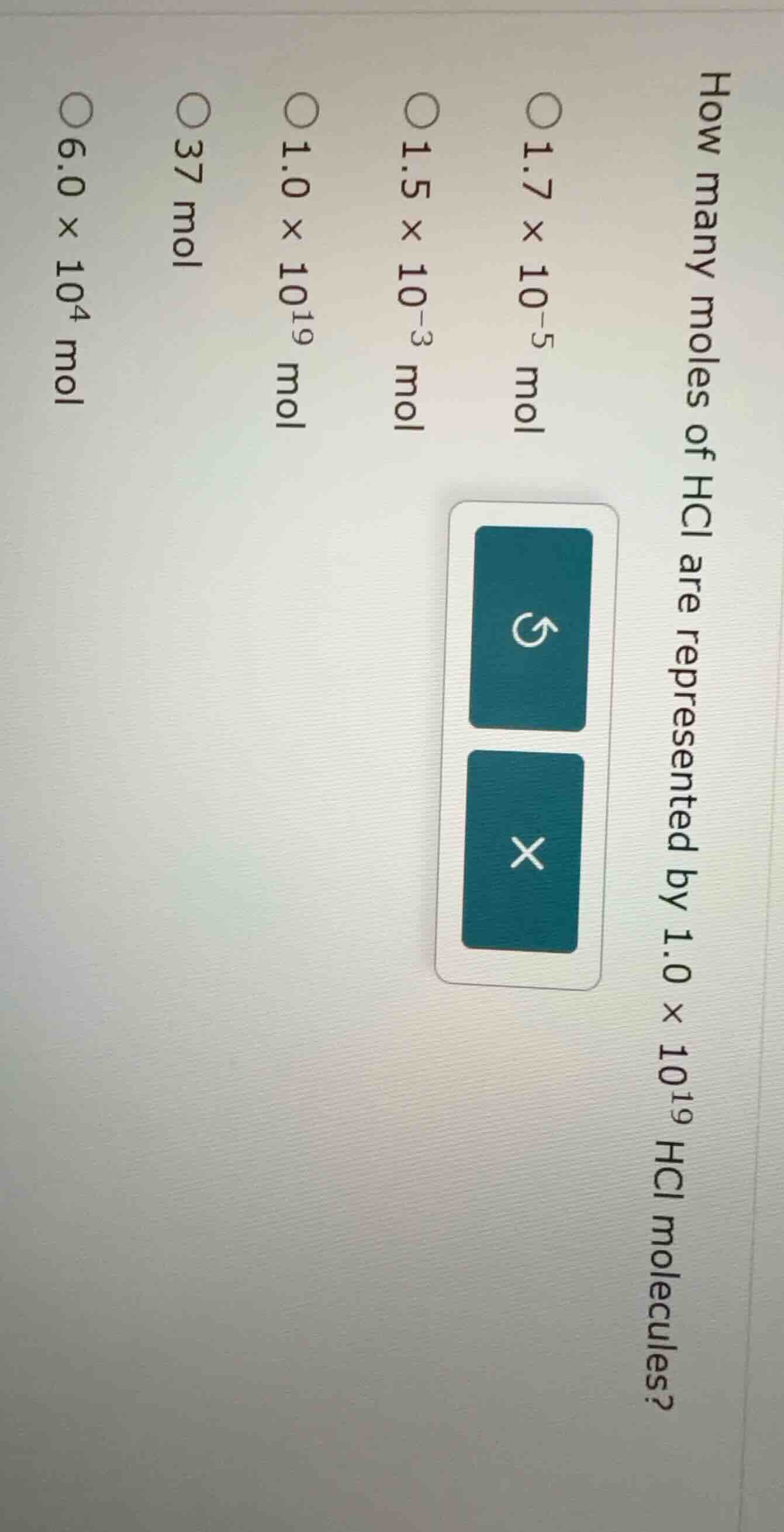
how many moles of hcl are represented by 1.0 × 10¹⁹ hcl molecules? 1.7 × 10⁻⁵ mol 1.5 × 10⁻³ mol 1.0 × 10¹⁹ mol 37 mol 6.0 × 10⁴ mol
Step1: Recall Avogadro's number
Avogadro's number is \( 6.022 \times 10^{23} \) molecules/mol, which relates the number of molecules to moles. The formula to find moles (\( n \)) from number of molecules (\( N \)) is \( n=\frac{N}{N_A} \), where \( N_A \) is Avogadro's number.
Step2: Substitute the values
Given \( N = 1.0\times 10^{19} \) molecules and \( N_A=6.022\times 10^{23} \) molecules/mol. So, \( n=\frac{1.0\times 10^{19}}{6.022\times 10^{23}} \)
Step3: Calculate the result
Simplify the expression: \( \frac{1.0\times 10^{19}}{6.022\times 10^{23}}=\frac{1.0}{6.022}\times 10^{19 - 23}\approx0.166\times 10^{-4}=1.66\times 10^{-5}\approx1.7\times 10^{-5} \) mol.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 1.7\times 10^{-5} \) mol (corresponding to the option: \( 1.7\times 10^{-5} \) mol)