QUESTION IMAGE
Question
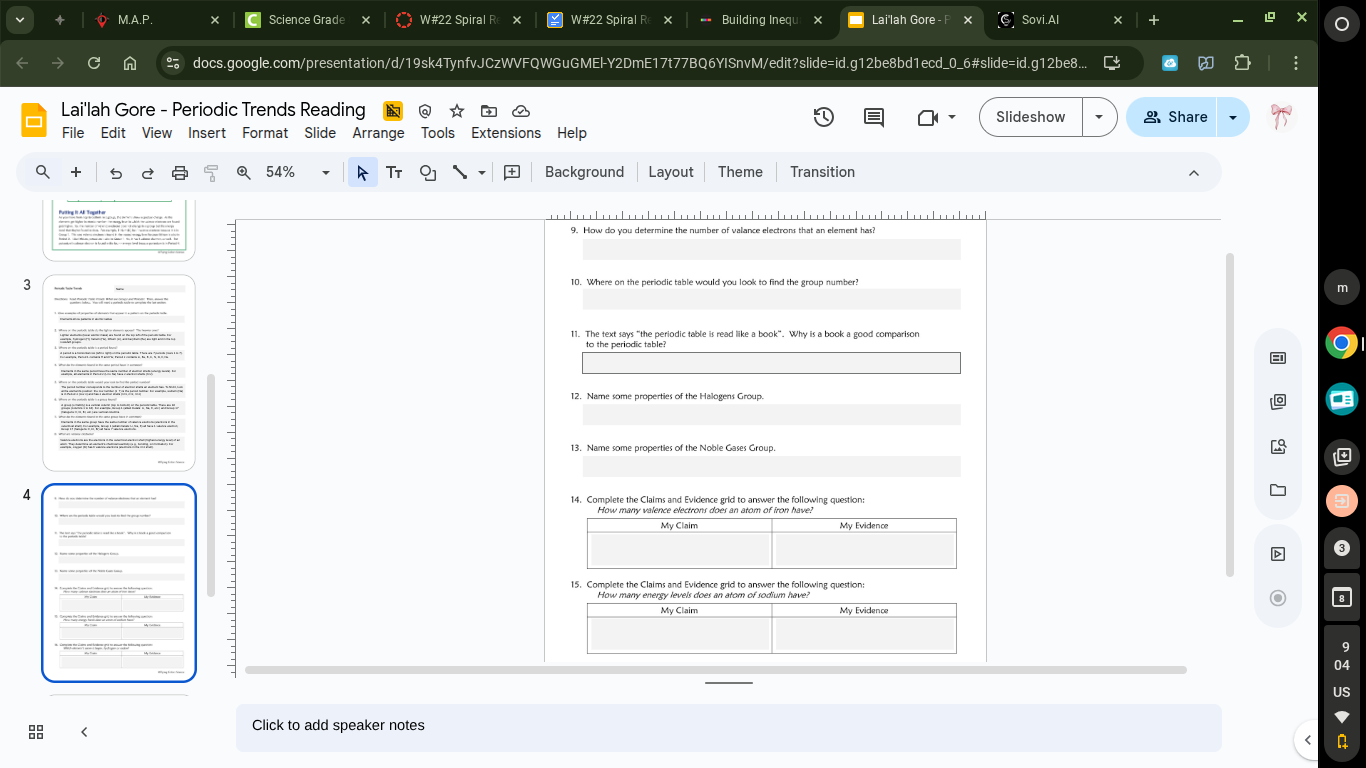
- how do you determine the number of valence electrons that an element has?
- where on the periodic table would you look to find the group number?
- the text says \the periodic table is like read a book\. why is a book a good comparison
to the periodic table?
- name some properties of the halogens group.
- name some properties of the noble gases group.
- complete the claims and evidence grid to answer the following question:
how many valence electrons does an atom of neon have?
my claim \tmy evidence
- complete the claims and evidence grid to answer the following question:
how many energy levels does an atom of calcium have?
my claim \tmy evidence
To answer these questions about periodic trends, we analyze each:
Question 9: Determine valence electrons
Valence electrons are determined by an element’s group number (column) on the periodic table. For main - group elements (Groups 1, 2, 13–18):
- Group 1: 1 valence electron.
- Group 2: 2 valence electrons.
- Groups 13–18: Valence electrons = group number - 10 (e.g., Group 14 has 4, Group 17 has 7).
Transition metals (Groups 3–12) have more variable valence electrons (based on electron configurations).
Question 10: Find group number
The group number (vertical column) is labeled at the top of each column on the periodic table. For example, Group 1 (alkali metals) is the first column, Group 18 (noble gases) is the last.
Question 11: Periodic table as a book
A book is a good comparison because:
- Like a book’s pages (organized sequentially), the periodic table’s rows (periods) are ordered by increasing atomic number (left - to - right, top - to - bottom).
- A book has chapters/sections; the periodic table has groups (columns) with elements sharing similar properties (e.g., Group 1 metals, Group 17 halogens).
- Just as a book’s structure helps find information, the periodic table’s organization (periods and groups) helps predict element properties.
Question 12: Halogens (Group 17) properties
- Physical: Non - metals, diatomic molecules (e.g., $\ce{Cl2}$, $\ce{Br2}$), exist as solids (iodine), liquids (bromine), or gases (fluorine, chlorine) at room temperature, colored (e.g., chlorine is yellow - green, bromine is red - brown).
- Chemical: Highly reactive non - metals, gain 1 electron to form $1^-$ ions, react vigorously with metals (e.g., $\ce{2Na + Cl2 -> 2NaCl}$), form salts (halides).
Question 13: Noble Gases (Group 18) properties
- Physical: Colorless, odorless gases at room temperature, very low boiling/melting points (monatomic molecules).
- Chemical: Extremely unreactive (inert) due to a full valence electron shell (8 electrons, except He with 2), rarely form compounds.
Question 14: Sodium (Na) valence electrons
- My Claim: A sodium atom has 1 valence electron.
- My Evidence: Sodium is in Group 1 of the periodic table. For main - group elements, the group number equals the number of valence electrons.
Question 15: Calcium (Ca) energy levels
- My Claim: A calcium atom has 4 energy levels (electron shells).
- My Evidence: Calcium is in Period 4 of the periodic table. The period number (horizontal row) equals the number of occupied energy levels (electron shells) in an atom.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s (summarized):
- Use the element’s group number (main - group: group # = valence e⁻; transition metals: variable).
- At the top of each vertical column (group).
- Organized by atomic number (rows = periods, columns = groups) like a book’s pages/sections.
- Halogens: reactive non - metals, diatomic, form $1^-$ ions, form salts.
- Noble Gases: inert, monatomic gases, full valence shell.
- Claim: 1 valence e⁻; Evidence: Group 1.
- Claim: 4 energy levels; Evidence: Period 4.