QUESTION IMAGE
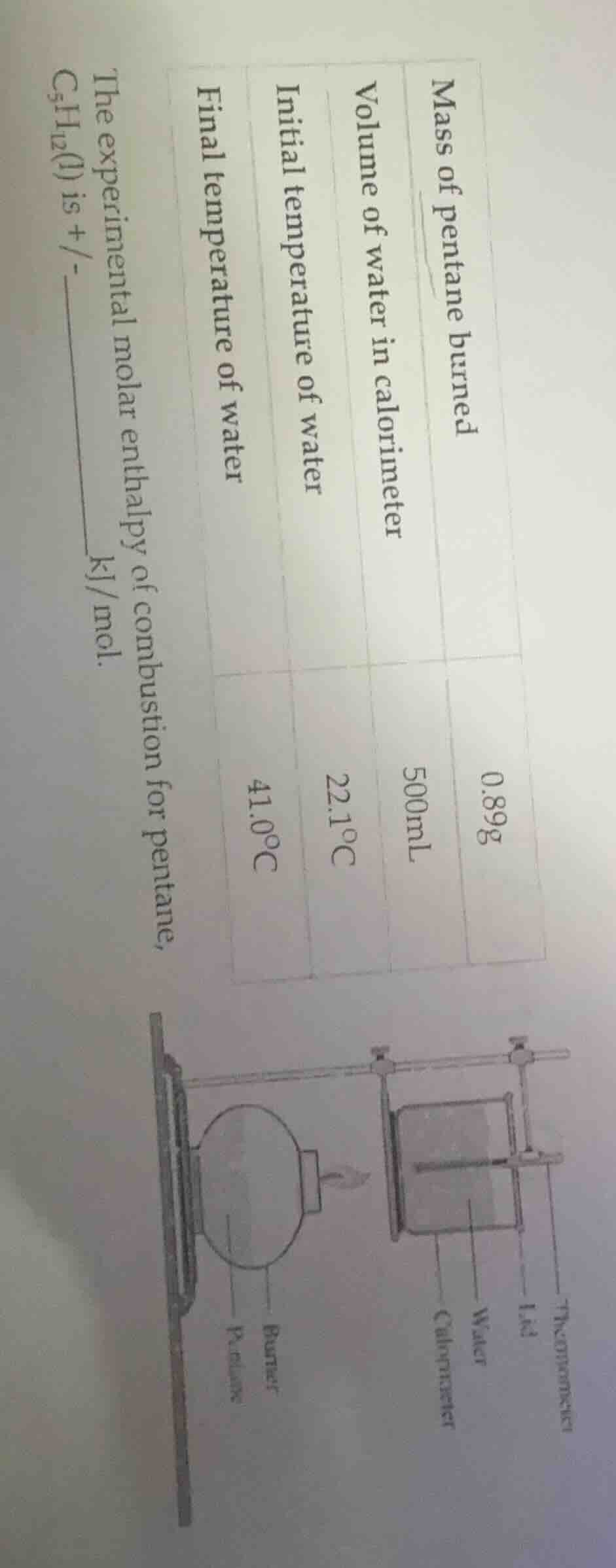
Question
Question was provided via image upload.
Step1: Calculate water mass
Since density of water is $1\ \text{g/mL}$, mass $m = 500\ \text{mL} \times 1\ \text{g/mL} = 500\ \text{g}$
Step2: Find temperature change
$\Delta T = T_{\text{final}} - T_{\text{initial}} = 41.0^\circ\text{C} - 22.1^\circ\text{C} = 18.9^\circ\text{C}$
Step3: Calculate heat absorbed by water
Use $q = mc\Delta T$, $c=4.184\ \text{J/g}^\circ\text{C}$:
$q = 500\ \text{g} \times 4.184\ \text{J/g}^\circ\text{C} \times 18.9^\circ\text{C} = 39550.8\ \text{J} = 39.5508\ \text{kJ}$
Step4: Moles of pentane burned
Molar mass of $\text{C}_5\text{H}_{12} = (5\times12.01)+(12\times1.008) = 72.15\ \text{g/mol}$
$n = \frac{0.89\ \text{g}}{72.15\ \text{g/mol}} \approx 0.01234\ \text{mol}$
Step5: Calculate molar enthalpy of combustion
Heat released by pentane = $-q$ (combustion is exothermic):
$\Delta H = -\frac{39.5508\ \text{kJ}}{0.01234\ \text{mol}} \approx -3200\ \text{kJ/mol}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{-3200}$ kJ/mol (rounded to 2 significant figures)