QUESTION IMAGE
Question
Question was provided via image upload.
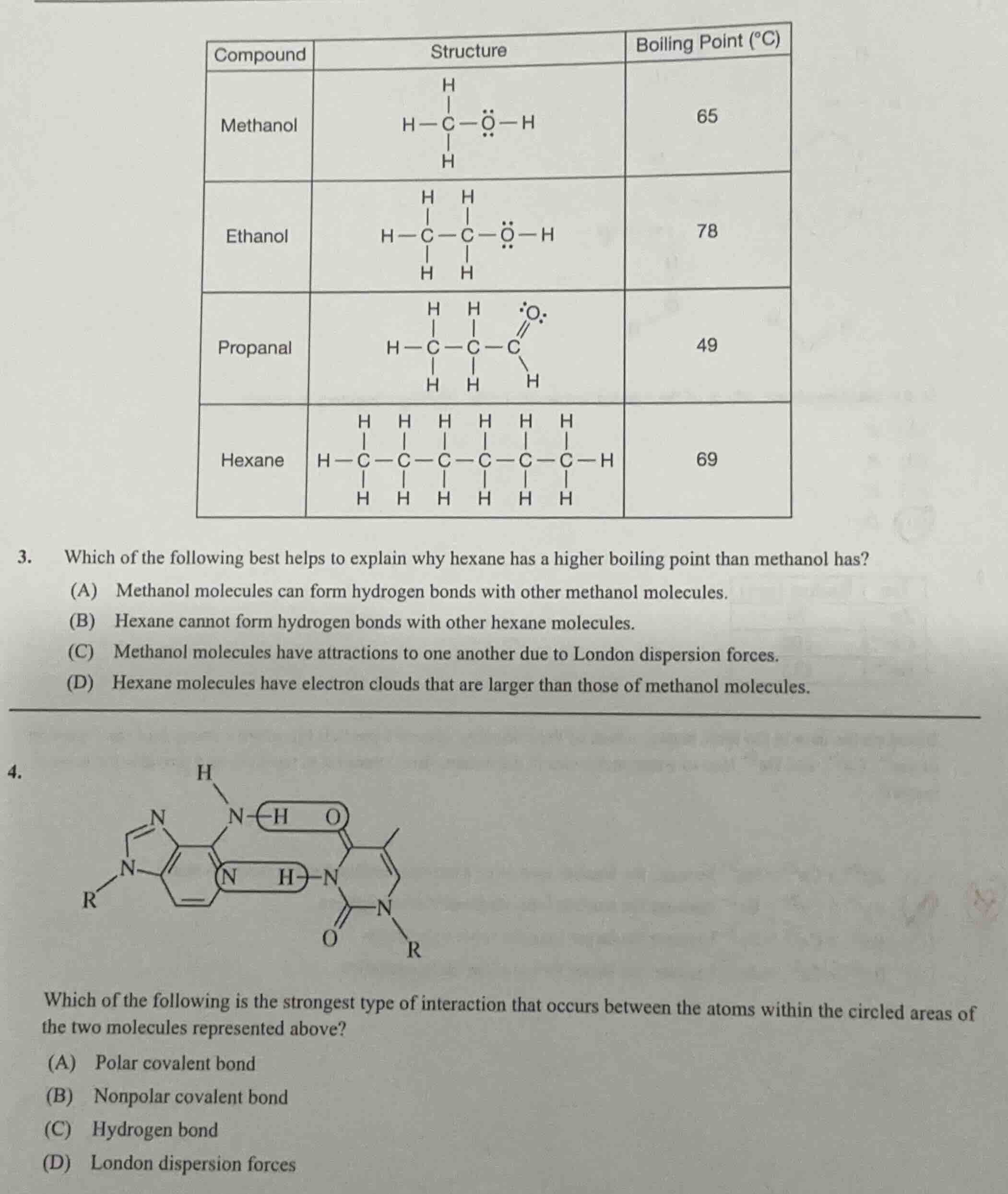
Question 3
Boiling points depend on intermolecular forces. Methanol has hydrogen bonding, a strong force, but hexane has a much larger electron cloud. Larger electron clouds create stronger London dispersion forces, and in this case, the cumulative dispersion forces in hexane outweigh the hydrogen bonding in methanol, leading to a higher boiling point. Option A only states methanol's hydrogen bonding, which would predict a higher boiling point for methanol, so it does not explain the trend. Option B only notes hexane lacks hydrogen bonding, which does not explain why its boiling point is higher. Option C is true but does not address the comparison to hexane's stronger forces. Option D correctly identifies that hexane's larger electron cloud leads to stronger dispersion forces that dominate over methanol's hydrogen bonding here.
The circled areas show hydrogen atoms bonded to highly electronegative nitrogen/oxygen atoms interacting with other electronegative nitrogen/oxygen atoms. Polar and nonpolar covalent bonds are intramolecular (within a single molecule), not intermolecular interactions between two molecules. London dispersion forces are weak, non-specific intermolecular forces, while hydrogen bonds are strong intermolecular interactions that form when a hydrogen is bonded to N, O, or F and interacts with another N, O, or F atom, which matches the circled interactions.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
(D) Hexane molecules have electron clouds that are larger than those of methanol molecules.
---