QUESTION IMAGE
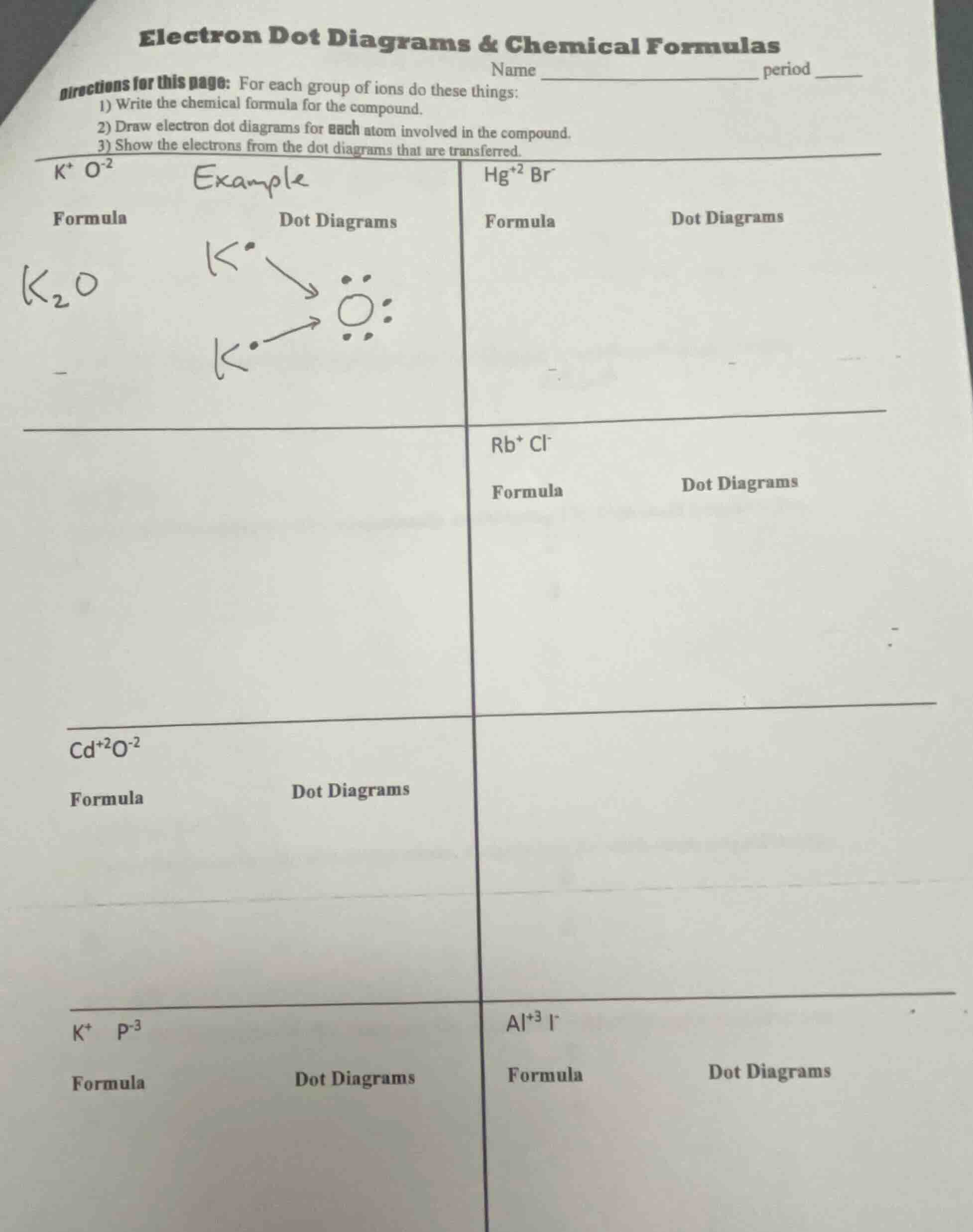
Question
Question was provided via image upload.
Step1: Balance charges for $\text{Hg}^{2+}\text{Br}^-$
To neutralize +2 charge of $\text{Hg}^{2+}$, 2 $\text{Br}^-$ are needed. Formula: $\text{HgBr}_2$. Hg has 2 valence electrons, each Br has 7; Hg transfers 1 electron to each Br.
Step2: Balance charges for $\text{Rb}^+\text{Cl}^-$
+1 and -1 charges cancel. Formula: $\text{RbCl}$. Rb has 1 valence electron, Cl has 7; Rb transfers 1 electron to Cl.
Step3: Balance charges for $\text{Cd}^{2+}\text{O}^{2-}$
+2 and -2 charges cancel. Formula: $\text{CdO}$. Cd has 2 valence electrons, O has 6; Cd transfers 2 electrons to O.
Step4: Balance charges for $\text{K}^+\text{P}^{3-}$
3 $\text{K}^+$ balance -3 charge of $\text{P}^{3-}$. Formula: $\text{K}_3\text{P}$. Each K has 1 valence electron, P has 5; each K transfers 1 electron to P.
Step5: Balance charges for $\text{Al}^{3+}\text{I}^-$
3 $\text{I}^-$ balance +3 charge of $\text{Al}^{3+}$. Formula: $\text{AlI}_3$. Al has 3 valence electrons, each I has 7; Al transfers 1 electron to each I.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\boldsymbol{HgBr_2}$; Electron dot diagram: $\text{Hg}^{\bullet\bullet}
ightarrow \overset{\Large \bullet\bullet}{\underset{\Large \bullet\bullet}{\text{Br}:}}$, $\text{Hg}^{\bullet\bullet}
ightarrow \overset{\Large \bullet\bullet}{\underset{\Large \bullet\bullet}{\text{Br}:}}$
- $\boldsymbol{RbCl}$; Electron dot diagram: $\text{Rb}^{\bullet}
ightarrow \overset{\Large \bullet\bullet}{\underset{\Large \bullet\bullet}{\text{Cl}:}}$
- $\boldsymbol{CdO}$; Electron dot diagram: $\text{Cd}^{\bullet\bullet}
ightarrow \overset{\Large \bullet\bullet}{\underset{\Large \bullet\bullet}{\text{O}:}}$
- $\boldsymbol{K_3P}$; Electron dot diagram: $\text{K}^{\bullet}
ightarrow \overset{\Large \bullet\bullet}{\underset{\Large \bullet\bullet}{\text{P}:}}$, $\text{K}^{\bullet}
ightarrow \overset{\Large \bullet\bullet}{\underset{\Large \bullet\bullet}{\text{P}:}}$, $\text{K}^{\bullet}
ightarrow \overset{\Large \bullet\bullet}{\underset{\Large \bullet\bullet}{\text{P}:}}$
- $\boldsymbol{AlI_3}$; Electron dot diagram: $\text{Al}^{\bullet\bullet\bullet}
ightarrow \overset{\Large \bullet\bullet}{\underset{\Large \bullet\bullet}{\text{I}:}}$, $\text{Al}^{\bullet\bullet\bullet}
ightarrow \overset{\Large \bullet\bullet}{\underset{\Large \bullet\bullet}{\text{I}:}}$, $\text{Al}^{\bullet\bullet\bullet}
ightarrow \overset{\Large \bullet\bullet}{\underset{\Large \bullet\bullet}{\text{I}:}}$