QUESTION IMAGE
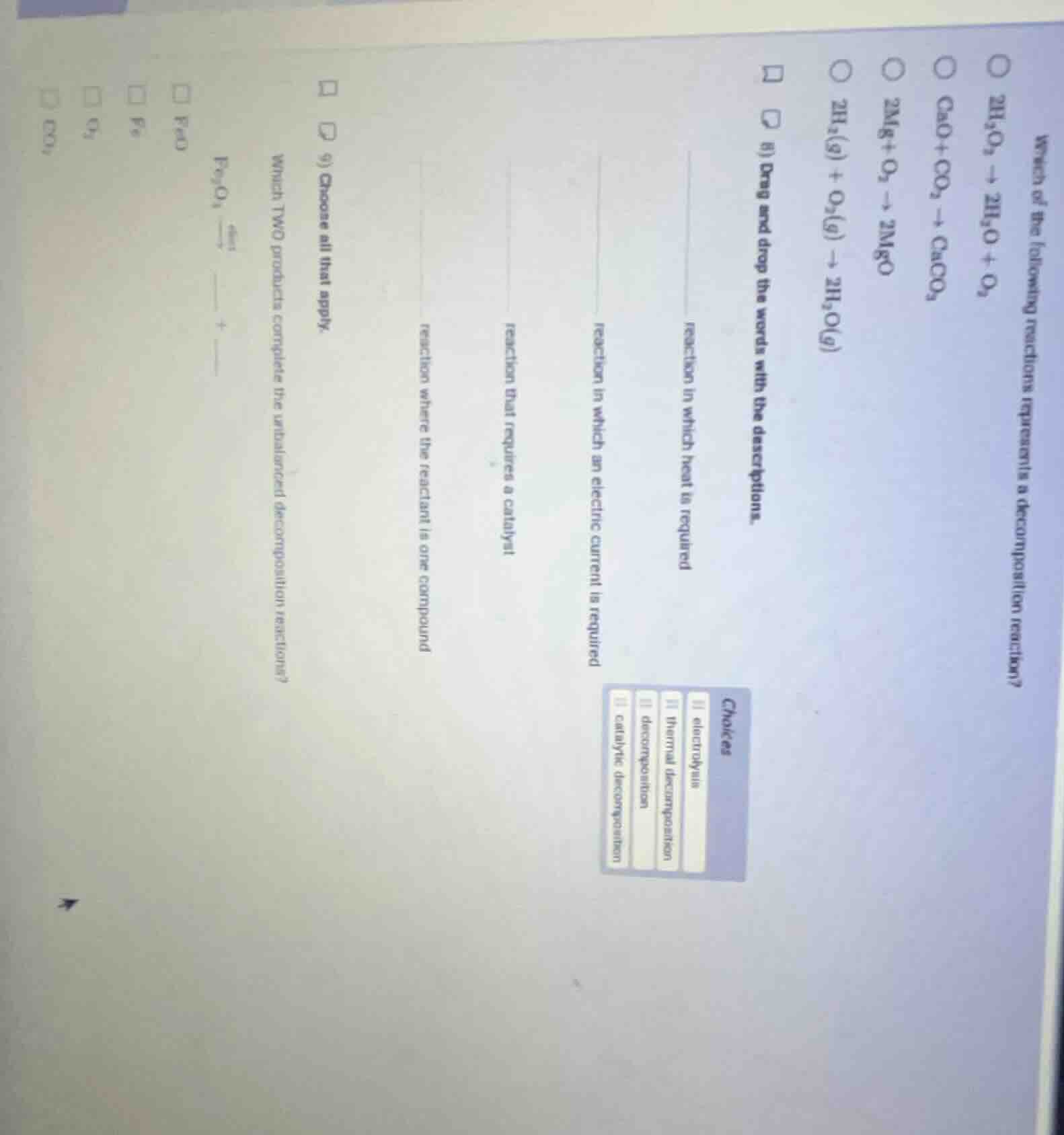
Question
Question was provided via image upload.
Step1: Identify decomposition reaction
A decomposition reaction has one compound breaking into simpler substances.
- $2H_2O_2
ightarrow 2H_2O + O_2$: 1 compound → 2 simpler substances
- $CaO+CO_2
ightarrow CaCO_3$: 2 reactants → 1 product (synthesis)
- $2Mg+O_2
ightarrow 2MgO$: 2 reactants → 1 product (synthesis)
- $2H_2(g) + O_2(g)
ightarrow 2H_2O(g)$: 2 reactants → 1 product (synthesis)
Step2: Match reaction descriptions
- Heat required: thermal decomposition
- Electric current required: electrolysis
- Catalyst required: catalytic decomposition
- Single compound reactant: decomposition
Step3: Complete Fe₂O₃ decomposition
Decomposition of $\text{Fe}_2\text{O}_3$ (heat) produces Fe and $\text{O}_2$:
$\text{Fe}_2\text{O}_3 \xrightarrow{\text{heat}} \text{Fe} + \text{O}_2$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Decomposition reaction: $\boldsymbol{2H_2O_2
ightarrow 2H_2O + O_2}$
- Drag-and-drop matches:
- reaction in which heat is required: thermal decomposition
- reaction in which an electric current is required: electrolysis
- reaction that requires a catalyst: catalytic decomposition
- reaction where the reactant is one compound: decomposition
- Completed decomposition: $\text{Fe}_2\text{O}_3 \xrightarrow{\text{heat}} \boldsymbol{\text{Fe}} + \boldsymbol{\text{O}_2}$ (selected products: Fe, $\text{O}_2$)